Diaphragm long-term facilitation following acute intermittent hypoxia during wakefulness and sleep
- PMID: 21372099
- PMCID: PMC3098661
- DOI: 10.1152/japplphysiol.00055.2011
Diaphragm long-term facilitation following acute intermittent hypoxia during wakefulness and sleep
Abstract
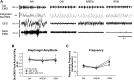
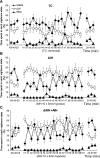
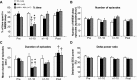
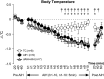
Acute intermittent hypoxia (AIH) elicits a form of respiratory plasticity known as long-term facilitation (LTF). Here, we tested four hypotheses in unanesthetized, spontaneously breathing rats using radiotelemetry for EEG and diaphragm electromyography (Dia EMG) activity: 1) AIH induces LTF in Dia EMG activity; 2) diaphragm LTF (Dia LTF) is more robust during sleep vs. wakefulness; 3) AIH (or repetitive AIH) disrupts natural sleep-wake architecture; and 4) preconditioning with daily AIH (dAIH) for 7 days enhances Dia LTF. Sleep-wake states and Dia EMG were monitored before (60 min), during, and after (60 min) AIH (10, 5-min hypoxic episodes, 5-min normoxic intervals; n = 9), time control (continuous normoxia, n = 8), and AIH following dAIH preconditioning for 7 days (n = 7). Dia EMG activities during quiet wakefulness (QW), rapid eye movement (REM), and non-REM (NREM) sleep were analyzed and normalized to pre-AIH values in the same state. During NREM sleep, diaphragm amplitude (25.1 ± 4.6%), frequency (16.4 ± 4.7%), and minute diaphragm activity (amplitude × frequency; 45.2 ± 6.6%) increased above baseline 0-60 min post-AIH (all P < 0.05). This Dia LTF was less robust during QW and insignificant during REM sleep. dAIH preconditioning had no effect on LTF (P > 0.05). We conclude that 1) AIH induces Dia LTF during NREM sleep and wakefulness; 2) Dia LTF is greater in NREM sleep vs. QW and is abolished during REM sleep; 3) AIH and repetitive AIH disrupt natural sleep patterns; and 4) Dia LTF is unaffected by dAIH. The capacity for plasticity in spinal pump muscles during sleep and wakefulness suggests an important role in the neural control of breathing.
Figures



References
-
- Aboubakr SE, Taylor A, Ford R, Siddiqi S, Badr MS. Long-term facilitation in obstructive sleep apnea patients during NREM sleep. J Appl Physiol 91: 2751–2757, 2001. - PubMed
-
- Babcock M, Shkoukani M, Aboubakr SE, Badr MS. Determinants of long-term facilitation in humans during NREM sleep. J Appl Physiol 94: 53–59, 2003. - PubMed
-
- Babcock MA, Badr MS. Long-term facilitation of ventilation in humans during NREM sleep. Sleep 21: 709–716, 1998. - PubMed
-
- Baker-Herman TL, Fuller DD, Bavis RW, Zabka AG, Golder FJ, Doperalski NJ, Johnson RA, Watters JJ, Mitchell GS. BDNF is necessary and sufficient for spinal respiratory plasticity following intermittent hypoxia. Nat Neurosci 7: 48–55, 2004. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

