Increased expression of interleukin-6 family members and receptors in urinary bladder with cyclophosphamide-induced bladder inflammation in female rats
- PMID: 21373362
- PMCID: PMC3044559
- DOI: 10.3389/fnins.2011.00020
Increased expression of interleukin-6 family members and receptors in urinary bladder with cyclophosphamide-induced bladder inflammation in female rats
Abstract
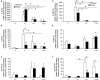
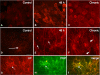
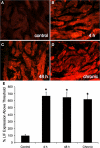
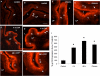
Recent studies suggest that janus-activated kinases-signal transducer and activator of transcription signaling pathways contribute to increased voiding frequency and referred pain of cyclophosphamide (CYP)-induced cystitis in rats. Potential upstream chemical mediator(s) that may be activated by CYP-induced cystitis to stimulate JAK/STAT signaling are not known in detail. In these studies, members of the interleukin (IL)-6 family of cytokines including, leukemia inhibitory factor (LIF), IL-6, and ciliary neurotrophic factor (CNTF) and associated receptors, IL-6 receptor (R) α, LIFR, and gp130 were examined in the urinary bladder in control and CYP-treated rats. Cytokine and receptor transcript and protein expression and distribution were determined in urinary bladder after CYP-induced cystitis using quantitative, real-time polymerase chain reaction (Q-PCR), western blotting, and immunohistochemistry. Acute (4 h; 150 mg/kg; i.p.), intermediate (48 h; 150 mg/kg; i.p.), or chronic (75 mg/kg; i.p., once every 3 days for 10 days) cystitis was induced in adult, female Wistar rats with CYP treatment. Q-PCR analyses revealed significant (p ≤ 0.01) CYP duration- and tissue- (e.g., urothelium, detrusor) dependent increases in LIF, IL-6, IL-6Rα, LIFR, and gp130 mRNA expression. Western blotting demonstrated significant (p ≤ 0.01) increases in IL-6, LIF, and gp130 protein expression in whole urinary bladder with CYP treatment. CYP-induced cystitis significantly (p ≤ 0.01) increased LIF-immunoreactivity (IR) in urothelium, detrusor, and suburothelial plexus whereas increased gp130-IR was only observed in urothelium and detrusor. These studies suggest that IL-6 and LIF may be potential upstream chemical mediators that activate JAK/STAT signaling in urinary bladder pathways.
Keywords: LIF; Q-PCR; Western blotting; detrusor smooth muscle; gp130; urothelium.
Figures


Similar articles
-
Phosphorylation of extracellular signal-regulated kinases in urinary bladder in rats with cyclophosphamide-induced cystitis.Am J Physiol Regul Integr Comp Physiol. 2007 Jul;293(1):R125-34. doi: 10.1152/ajpregu.00857.2006. Epub 2007 Apr 4. Am J Physiol Regul Integr Comp Physiol. 2007. PMID: 17409261
-
Expression of corticotropin-releasing factor and CRF receptors in micturition pathways after cyclophosphamide-induced cystitis.Am J Physiol Regul Integr Comp Physiol. 2006 Sep;291(3):R692-703. doi: 10.1152/ajpregu.00086.2006. Epub 2006 Apr 13. Am J Physiol Regul Integr Comp Physiol. 2006. PMID: 16614059
-
Expression and response of acid-sensing ion channels in urinary bladder to cyclophosphamide-induced cystitis.Am J Physiol Renal Physiol. 2010 May;298(5):F1130-9. doi: 10.1152/ajprenal.00618.2009. Epub 2010 Feb 17. Am J Physiol Renal Physiol. 2010. PMID: 20164155 Free PMC article.
-
Expression of cyclooxygenase-2 in urinary bladder in rats with cyclophosphamide-induced cystitis.Am J Physiol Regul Integr Comp Physiol. 2007 Aug;293(2):R677-85. doi: 10.1152/ajpregu.00305.2007. Epub 2007 May 30. Am J Physiol Regul Integr Comp Physiol. 2007. PMID: 17537839
-
The tripartite CNTF receptor complex: activation and signaling involves components shared with other cytokines.J Neurobiol. 1994 Nov;25(11):1454-66. doi: 10.1002/neu.480251111. J Neurobiol. 1994. PMID: 7852997 Review.
Cited by
-
Expression and function of transforming growth factor-β isoforms and cognate receptors in the rat urinary bladder following cyclophosphamide-induced cystitis.Am J Physiol Renal Physiol. 2013 Nov 1;305(9):F1265-76. doi: 10.1152/ajprenal.00042.2013. Epub 2013 Aug 7. Am J Physiol Renal Physiol. 2013. PMID: 23926183 Free PMC article.
-
Suppression of the PI3K pathway in vivo reduces cystitis-induced bladder hypertrophy and restores bladder capacity examined by magnetic resonance imaging.PLoS One. 2014 Dec 8;9(12):e114536. doi: 10.1371/journal.pone.0114536. eCollection 2014. PLoS One. 2014. PMID: 25486122 Free PMC article.
-
Transient Effects of Cyclophosphamide on Basal Cell Proliferation of Olfactory Epithelia.Chem Senses. 2020 Oct 9;45(7):549-561. doi: 10.1093/chemse/bjaa039. Chem Senses. 2020. PMID: 32531016 Free PMC article.
-
microRNA-146a Promotes Growth of Acute Leukemia Cells by Downregulating Ciliary Neurotrophic Factor Receptor and Activating JAK2/STAT3 Signaling.Yonsei Med J. 2019 Oct;60(10):924-934. doi: 10.3349/ymj.2019.60.10.924. Yonsei Med J. 2019. PMID: 31538427 Free PMC article.
-
Detrusor myocyte autophagy protects the bladder function via inhibiting the inflammation in cyclophosphamide-induced cystitis in rats.PLoS One. 2015 Apr 1;10(4):e0122597. doi: 10.1371/journal.pone.0122597. eCollection 2015. PLoS One. 2015. PMID: 25830308 Free PMC article.
References
-
- Abramoff M. D., Magelhaes P. J., Ram S. J. (2004). Image processing with ImageJ. Biophotonics Int. 11, 36–42
-
- Andersson K. E., Wein A. J. (2004). Pharmacology of the lower urinary tract: basis for current and future treatments of urinary incontinence. Pharmacol. Rev. 56, 581–631 - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

