Clinically translatable cell tracking and quantification by MRI in cartilage repair using superparamagnetic iron oxides
- PMID: 21373640
- PMCID: PMC3044153
- DOI: 10.1371/journal.pone.0017001
Clinically translatable cell tracking and quantification by MRI in cartilage repair using superparamagnetic iron oxides
Abstract
Background: Articular cartilage has very limited intrinsic regenerative capacity, making cell-based therapy a tempting approach for cartilage repair. Cell tracking can be a major step towards unraveling and improving the repair process of these therapies. We studied superparamagnetic iron oxides (SPIO) for labeling human bone marrow-derived mesenchymal stem cells (hBMSCs) regarding effectivity, cell viability, long term metabolic cell activity, chondrogenic differentiation and hBMSC secretion profile. We additionally examined the capacity of synovial cells to endocytose SPIO from dead, labeled cells, together with the use of magnetic resonance imaging (MRI) for intra-articular visualization and quantification of SPIO labeled cells.
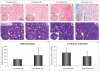
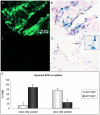
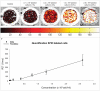
Methodology/prinicipal findings: Efficacy and various safety aspects of SPIO cell labeling were determined using appropriate assays. Synovial SPIO re-uptake was investigated in vitro by co-labeling cells with SPIO and green fluorescent protein (GFP). MRI experiments were performed on a clinical 3.0T MRI scanner. Two cell-based cartilage repair techniques were mimicked for evaluating MRI traceability of labeled cells: intra-articular cell injection and cell implantation in cartilage defects. Cells were applied ex vivo or in vitro in an intra-articular environment and immediately scanned. SPIO labeling was effective and did not impair any of the studied safety aspects, including hBMSC secretion profile. SPIO from dead, labeled cells could be taken up by synovial cells. Both injected and implanted SPIO-labeled cells could accurately be visualized by MRI in a clinically relevant sized joint model using clinically applied cell doses. Finally, we quantified the amount of labeled cells seeded in cartilage defects using MR-based relaxometry.
Conclusions: SPIO labeling appears to be safe without influencing cell behavior. SPIO labeled cells can be visualized in an intra-articular environment and quantified when seeded in cartilage defects.
Conflict of interest statement
Figures


References
-
- Brittberg M, Lindahl A, Nilsson A, Ohlsson C, Isaksson O, et al. Treatment of deep cartilage defects in the knee with autologous chondrocyte transplantation. N Engl J Med. 1994;331:889–895. - PubMed
-
- Centeno CJ, Schultz JR, Cheever M, Robinson B, Freeman M, et al. Safety and complications reporting on the re-implantation of culture-expanded mesenchymal stem cells using autologous platelet lysate technique. Curr Stem Cell Res Ther. 2010;5:81–93. - PubMed
-
- Wakitani S, Nawata M, Tensho K, Okabe T, Machida H, et al. Repair of articular cartilage defects in the patello-femoral joint with autologous bone marrow mesenchymal cell transplantation: three case reports involving nine defects in five knees. J Tissue Eng Regen Med. 2007;1:74–79. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

