Effects of morphine on pain-elicited and pain-suppressed behavior in CB1 knockout and wildtype mice
- PMID: 21373789
- PMCID: PMC3160632
- DOI: 10.1007/s00213-011-2232-5
Effects of morphine on pain-elicited and pain-suppressed behavior in CB1 knockout and wildtype mice
Abstract
Rationale: Pharmacological manipulations of the type 1 cannabinoid receptor (CB1) suggest a role for CB1 in morphine-induced antinociception, but studies utilizing CB1 knockout (KO) mice do not support this conclusion. Since studies using CB1 KO mice to study morphine's antinociceptive effects have only examined thermal nociception, this study examines these interactions in models that employ a chemical stimulus.
Objectives: To determine whether the findings obtained with thermal pain models extend to other models, the effects of morphine on acetic acid-induced writhing were examined in CB1 KO and wildtype (WT) mice. Behaviors that decrease in response to acid injection, feeding and wheel running, were also examined, and investigations were carried out in the thermal hotplate assay. The CB1 antagonist SR141716A was also examined in these assays.
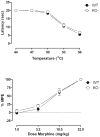
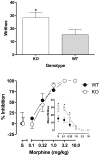
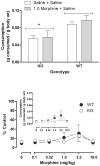
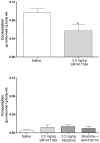
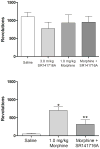
Results: Morphine completely blocked acid-induced writhing (1.0-10.0 mg/kg) and increased response latencies in the hotplate (10.0-32.0 mg/kg) in both genotypes. Morphine (3.2 mg/kg) significantly attenuated the suppression of wheel running but did not completely prevent this effect in either genotype. Morphine did not alter pain-suppressed feeding. In each of these assays, morphine's effects were not altered in CB1 KO mice compared with WT mice; however, SR141716A attenuated morphine's effects in C57BL/6 mice.
Conclusions: The effects of morphine do not differ in CB1 KO and WT mice in preclinical pain models using thermal and chemical stimuli. Since SR141716A did attenuate the effects of morphine, it is possible that CB1 KO mice undergo developmental changes that mask the role of CB1 receptors in morphine's antinociceptive effects.
Figures



References
-
- Ates M, Hamza M, Seidel K, Kotalla CE, Ledent C, Guhring H. Intrathecally applied flurbiprofren produces an endocannabinoid-dependent antinociception in the rat formalin test. Eur J Neurosci. 2003;17:597–604. - PubMed
-
- Calignano A, La Rana G, Giuffrida A, Piomelli D. Control of pain by endogenous cannabinoids. Nature. 1998;394:277–281. - PubMed
-
- Cossu G, Ledent C, Fattore L, Imperato A, Bohme GA, Parmentier M, Fratta W. Cannabinoid CB1 receptor knockout mice fail to self-administer morphine but not other drugs of abuse. Behav Brain Res. 2001;118:61–65. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

