Cannabinoid 1 receptor activation contributes to vascular inflammation and cell death in a mouse model of diabetic retinopathy and a human retinal cell line
- PMID: 21373835
- PMCID: PMC3375271
- DOI: 10.1007/s00125-011-2061-4
Cannabinoid 1 receptor activation contributes to vascular inflammation and cell death in a mouse model of diabetic retinopathy and a human retinal cell line
Abstract
Aims/hypothesis: Recent studies have demonstrated that cannabinoid-1 (CB(1)) receptor blockade ameliorated inflammation, endothelial and/or cardiac dysfunction, and cell death in models of nephropathy, atherosclerosis and cardiomyopathy. However the role of CB(1) receptor signalling in diabetic retinopathy remains unexplored. Using genetic deletion or pharmacological inhibition of the CB(1) receptor with SR141716 (rimonabant) in a rodent model of diabetic retinopathy or in human primary retinal endothelial cells (HREC) exposed to high glucose, we explored the role of CB(1) receptors in the pathogenesis of diabetic retinopathy.
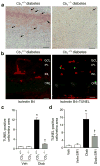
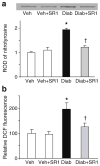
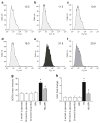
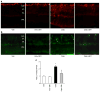
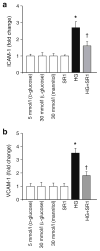
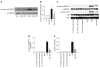
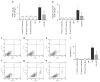
Methods: Diabetes was induced using streptozotocin in C57BL/6J Cb(1) (also known as Cnr1)(+/+) and Cb(1)(-/-) mice aged 8 to 12 weeks. Samples from mice retina or HREC were used to determine: (1) apoptosis; (2) activity of nuclear factor kappa B, intercellular adhesion molecule 1 (ICAM-1), vascular cell adhesion molecule 1 (VCAM-1), poly (ADP-ribose) polymerase and caspase-3; (3) content of 3-nitrotyrosine and reactive oxygen species; and (4) activation of p38/Jun N-terminal kinase/mitogen-activated protein kinase (MAPK).
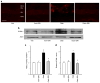
Results: Deletion of CB(1) receptor or treatment of diabetic mice with CB(1) receptor antagonist SR141716 prevented retinal cell death. Treatment of diabetic mice or HREC cells exposed to high glucose with SR141716 attenuated the oxidative and nitrative stress, and reduced levels of nuclear factor κB, ICAM-1 and VCAM-1. In addition, SR141716 attenuated the diabetes- or high glucose-induced pro-apoptotic activation of MAPK and retinal vascular cell death.
Conclusions/interpretation: Activation of CB(1) receptors may play an important role in the pathogenesis of diabetic retinopathy by facilitating MAPK activation, oxidative stress and inflammatory signalling. Conversely, CB(1) receptor inhibition may be beneficial in the treatment of this devastating complication of diabetes.
Conflict of interest statement
Figures
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

