Disruption of downstream MyD88 or TRIF Toll-like receptor signaling does not protect against cerebral ischemia
- PMID: 21376021
- PMCID: PMC3126100
- DOI: 10.1016/j.brainres.2011.02.074
Disruption of downstream MyD88 or TRIF Toll-like receptor signaling does not protect against cerebral ischemia
Abstract
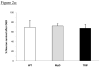
Toll-like receptor (TLR) signaling plays an important role in cerebral ischemia, but downstream signaling events, which can be organ-specific, are incompletely understood. We thereby investigated involvement of the MyD88-dependent (MyD88) and MyD88-independent (TRIF) TLR signaling pathways in 2 in vitro and 2 in vivo models of cerebral ischemia. For in vitro studies, we used a model of oxygen-glucose deprivation (OGD) followed by flow cytometric analysis to determine:1) viability of PC12 cells following knock-down with MyD88 siRNA compared to negative control siRNA and 2) viability, apoptosis and necrosis of cortical neurons from MyD88 null (-/-) , TRIF mutant, and wild type (WT) mice. In addition, in vivo, 1) We examined CA1 neuronal survival 7 days after global forebrain ischemia and 2) infarct volumes 24h after Middle Cerebral Artery Occlusion (MCAO) in all 3 types of mice. OGD: 1) There were no differences in either percent viability of PC12 cells transfected with MyD88 compared to negative control siRNA or 2) in percent viability, apoptosis and necrosis of cortical neurons from MyD88-/-,TRIF mutant and WT mice. Global ischemia: neuronal survival was similar in all 3 groups of mice. Finally, MCAO: infarct volumes were not statistically different among all 3 groups of mice: MyD88-/-, 23.9±9.9 mm(3), TRIF mutant, 26.7±5.8 mm(3) and WT, 17.9±8.4mm(3). These findings show that disruption of MyD88 or TRIF signaling does not confer protection in brain ischemia and suggests the possibility of additional or alternate downstream adaptors during TLR signaling in cerebral ischemia.
Published by Elsevier B.V.
Conflict of interest statement
Figures








Similar articles
-
Downstream Toll-like receptor signaling mediates adaptor-specific cytokine expression following focal cerebral ischemia.J Neuroinflammation. 2012 Jul 16;9:174. doi: 10.1186/1742-2094-9-174. J Neuroinflammation. 2012. PMID: 22799573 Free PMC article.
-
TLR4-HMGB1-, MyD88- and TRIF-dependent signaling in mouse intestinal ischemia/reperfusion injury.World J Gastroenterol. 2015 Jul 21;21(27):8314-25. doi: 10.3748/wjg.v21.i27.8314. World J Gastroenterol. 2015. PMID: 26217083 Free PMC article.
-
A new role for downstream Toll-like receptor signaling in mediating immediate early gene expression during focal cerebral ischemia.J Cereb Blood Flow Metab. 2014 Feb;34(2):258-67. doi: 10.1038/jcbfm.2013.182. Epub 2013 Dec 4. J Cereb Blood Flow Metab. 2014. PMID: 24301291 Free PMC article.
-
The TIR-domain-containing adapter inducing interferon-β-dependent signaling cascade plays a crucial role in ischemia-reperfusion-induced retinal injury, whereas the contribution of the myeloid differentiation primary response 88-dependent signaling cascade is not as pivotal.Eur J Neurosci. 2014 Aug;40(3):2502-12. doi: 10.1111/ejn.12603. Epub 2014 Apr 23. Eur J Neurosci. 2014. PMID: 24754835 Free PMC article.
-
TLR5 expression in the small intestine depends on the adaptors MyD88 and TRIF, but is independent of the enteric microbiota.Gut Microbes. 2015;6(3):202-6. doi: 10.1080/19490976.2015.1034417. Epub 2015 Apr 29. Gut Microbes. 2015. PMID: 25923903 Free PMC article. Review.
Cited by
-
Poly (I:C) therapy decreases cerebral ischaemia/reperfusion injury via TLR3-mediated prevention of Fas/FADD interaction.J Cell Mol Med. 2015 Mar;19(3):555-65. doi: 10.1111/jcmm.12456. Epub 2014 Oct 29. J Cell Mol Med. 2015. PMID: 25351293 Free PMC article.
-
Toll-like receptor-3 activation increases the vulnerability of the neonatal brain to hypoxia-ischemia.J Neurosci. 2013 Jul 17;33(29):12041-51. doi: 10.1523/JNEUROSCI.0673-13.2013. J Neurosci. 2013. PMID: 23864690 Free PMC article.
-
Retinal dendritic cell recruitment, but not function, was inhibited in MyD88 and TRIF deficient mice.J Neuroinflammation. 2014 Aug 13;11:143. doi: 10.1186/s12974-014-0143-1. J Neuroinflammation. 2014. PMID: 25116321 Free PMC article.
-
Discovery of Intermediary Genes between Pathways Using Sparse Regression.PLoS One. 2015 Sep 8;10(9):e0137222. doi: 10.1371/journal.pone.0137222. eCollection 2015. PLoS One. 2015. PMID: 26348038 Free PMC article.
-
Downstream Toll-like receptor signaling mediates adaptor-specific cytokine expression following focal cerebral ischemia.J Neuroinflammation. 2012 Jul 16;9:174. doi: 10.1186/1742-2094-9-174. J Neuroinflammation. 2012. PMID: 22799573 Free PMC article.
References
-
- Adachi O, Kawai T, Takeda K, Matsumoto M, Tsutsui H, Sakagami M, Nakanishi K, Akira S. Targeted disruption of the MyD88 gene results in loss of IL-1- and IL-18-mediated function. Immunity. 1998;9:143–50. - PubMed
-
- Caso JR, Pradillo JM, Hurtado O, Lorenzo P, Moro MA, Lizasoain I. Toll-like receptor 4 is involved in brain damage and inflammation after experimental stroke. Circulation. 2007;115:1599–608. - PubMed
-
- Hua F, Ha T, Ma J, Gao X, Kelley J, Williams DL, Browder IW, Kao RL, Li C. Blocking the MyD88-dependent pathway protects the myocardium from ischemia/reperfusion injury in rat hearts. Biochem Biophys Res Commun. 2005;338:1118–25. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

