The anti-inflammatory effects of dimethyl fumarate in astrocytes involve glutathione and haem oxygenase-1
- PMID: 21382015
- PMCID: PMC3072764
- DOI: 10.1042/AN20100033
The anti-inflammatory effects of dimethyl fumarate in astrocytes involve glutathione and haem oxygenase-1
Abstract
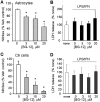
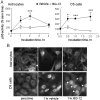
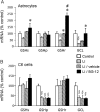
DMF (dimethyl fumarate) exerts anti-inflammatory and pro-metabolic effects in a variety of cell types, and a formulation (BG-12) is being evaluated for monotherapy in multiple sclerosis patients. DMF modifies glutathione (GSH) levels that can induce expression of the anti-inflammatory protein HO-1 (haem oxygenase-1). In primary astrocytes and C6 glioma cells, BG-12 dose-dependently suppressed nitrite production induced by either LI [LPS (lipopolysaccharide) at 1 μg/ml plus IFNγ (interferon γ) at 20 units/ml] or a mixture of pro-inflammatory cytokines, with greater efficacy in C6 cells. BG-12 reduced NOS2 (nitric oxide synthase 2) mRNA levels and activation of a NOS2 promoter, reduced nuclear levels of NF-κB (nuclear factor κB) p65 subunit and attenuated loss of IκBα (inhibitory κBα) in both cell types, although with greater effects in astrocytes. In astrocytes, LI decreased mRNA levels for GSHr (GSH reductase) and GCL (c-glutamylcysteine synthetase), and slightly suppressed GSHs (GSH synthetase) mRNAs. Co-treatment with BG-12 prevented those decreased and increased levels above control values. In contrast, LI reduced GSHp (GSH peroxidase) and GCL in C6 cells, and BG-12 had no effect on those levels. BG-12 increased nuclear levels of Nrf2 (nuclear factor-erythroid 2 p45 subunit-related factor 2), an inducer of GSH-related enzymes, in astrocytes but not C6 cells. In astrocytes, GSH was decreased by BG-12 at 2 h and increased at 24 h. Prior depletion of GSH using buthionine-sulfoximine increased the ability of BG-12 to reduce nitrites. In astrocytes, BG-12 increased HO-1 mRNA levels and effects on nitrite levels were blocked by an HO-1 inhibitor. These results demonstrate that BG-12 suppresses inflammatory activation in astrocytes and C6 glioma cells, but with distinct mechanisms, different dependence on GSH and different effects on transcription factor activation.
Figures
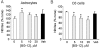
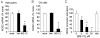

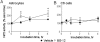
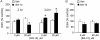
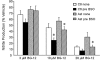
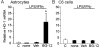
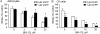
Similar articles
-
Dimethyl fumarate and monoethyl fumarate exhibit differential effects on KEAP1, NRF2 activation, and glutathione depletion in vitro.PLoS One. 2015 Mar 20;10(3):e0120254. doi: 10.1371/journal.pone.0120254. eCollection 2015. PLoS One. 2015. PMID: 25793262 Free PMC article.
-
Dimethyl Fumarate Attenuates Di-(2-Ethylhexyl) Phthalate-Induced Nephrotoxicity Through the Nrf2/HO-1 and NF-κB Signaling Pathways.Inflammation. 2023 Feb;46(1):453-467. doi: 10.1007/s10753-022-01746-6. Epub 2022 Oct 5. Inflammation. 2023. PMID: 36195817
-
Lipoxin A4 Activates Nrf2 Pathway and Ameliorates Cell Damage in Cultured Cortical Astrocytes Exposed to Oxygen-Glucose Deprivation/Reperfusion Insults.J Mol Neurosci. 2015 Aug;56(4):848-857. doi: 10.1007/s12031-015-0525-6. Epub 2015 Feb 22. J Mol Neurosci. 2015. PMID: 25702137
-
2'-Methoxy-4'6'-bis(methoxymethoxy)chalcone inhibits nitric oxide production in lipopolysaccharide-stimulated RAW 264.7 macrophages.Basic Clin Pharmacol Toxicol. 2010 Jun;106(6):454-60. doi: 10.1111/j.1742-7843.2009.00524.x. Epub 2010 Jan 18. Basic Clin Pharmacol Toxicol. 2010. PMID: 20088848
-
Dimethyl fumarate: A review of preclinical efficacy in models of neurodegenerative diseases.Eur J Pharmacol. 2022 Jul 5;926:175025. doi: 10.1016/j.ejphar.2022.175025. Epub 2022 May 13. Eur J Pharmacol. 2022. PMID: 35569547 Review.
Cited by
-
Treatment-Related Progressive Multifocal Leukoencephalopathy in Multiple Sclerosis: A Comprehensive Review of Current Evidence and Future Needs.Drug Saf. 2016 Dec;39(12):1163-1174. doi: 10.1007/s40264-016-0461-6. Drug Saf. 2016. PMID: 27696299 Review.
-
The Effects of Dimethyl Fumarate on Atherosclerosis in the Apolipoprotein E-Deficient Mouse Model with Streptozotocin-Induced Hyperglycemia Mediated By the Nuclear Factor Erythroid 2-Related Factor 2/Antioxidant Response Element (Nrf2/ARE) Signaling Pathway.Med Sci Monit. 2019 Oct 24;25:7966-7975. doi: 10.12659/MSM.918951. Med Sci Monit. 2019. PMID: 31645538 Free PMC article.
-
Dimethyl fumarate modulation of immune and antioxidant responses: application to HIV therapy.Crit Rev Immunol. 2013;33(4):307-59. doi: 10.1615/critrevimmunol.2013007247. Crit Rev Immunol. 2013. PMID: 23971529 Free PMC article. Review.
-
Treatment with dimethyl fumarate reduces the formation and rupture of intracranial aneurysms: Role of Nrf2 activation.J Cereb Blood Flow Metab. 2020 May;40(5):1077-1089. doi: 10.1177/0271678X19858888. Epub 2019 Jun 20. J Cereb Blood Flow Metab. 2020. PMID: 31220996 Free PMC article.
-
The Evolving Role of Monomethyl Fumarate Treatment as Pharmacotherapy for Relapsing-Remitting Multiple Sclerosis.Cureus. 2024 Apr 6;16(4):e57714. doi: 10.7759/cureus.57714. eCollection 2024 Apr. Cureus. 2024. PMID: 38711693 Free PMC article. Review.
References
-
- Brenner T, Brocke S, Szafer F, Sobel RA, Parkinson JF, Perez DH, Steinman L. Inhibition of nitric oxide synthase for treatment of experimental autoimmune encephalomyelitis. J Immunol. 1997;158:2940–2946. - PubMed
-
- Broholm H, Andersen B, Wanscher B, Frederiksen JL, Rubin I, Pakkenberg B, Larsson HB, Lauritzen M. Nitric oxide synthase expression and enzymatic activity in multiple sclerosis. Acta Neurol Scand. 2004;109:261–269. - PubMed
-
- Brosnan CF, Battistini L, Raine CS, Dickson DW, Casadevall A, Lee SC. Reactive nitrogen intermediates in human neuropathology: an overview. Dev Neurosci. 1994;16:152–161. - PubMed
-
- Calabrese V, Ravagna A, Colombrita C, Scapagnini G, Guagliano E, Calvani M, Butterfield DA, Giuffrida Stella AM. Acetylcarnitine induces heme oxygenase in rat astrocytes and protects against oxidative stress: involvement of the transcription factor Nrf2. J Neurosci Res. 2005;79:509–521. - PubMed
-
- Dethlefsen LA, Lehman CM, Biaglow JE, Peck VM. Toxic effects of acute glutathione depletion by buthionine sulfoximine and dimethylfumarate on murine mammary carcinoma cells. Radiat Res. 1988;114:215–224. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

