Isoform-specific regulation of the inositol 1,4,5-trisphosphate receptor by O-linked glycosylation
- PMID: 21383013
- PMCID: PMC3091177
- DOI: 10.1074/jbc.M110.206482
Isoform-specific regulation of the inositol 1,4,5-trisphosphate receptor by O-linked glycosylation
Abstract
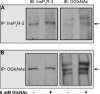
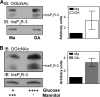
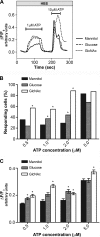
The inositol 1,4,5-trisphosphate receptor (InsP(3)R), an intracellular calcium channel, has three isoforms with >65% sequence homology, yet the isoforms differ in their function and regulation by post-translational modifications. We showed previously that InsP(3)R-1 is functionally modified by O-linked β-N-acetylglucosamine glycosylation (O-GlcNAcylation) (Rengifo, J., Gibson, C. J., Winkler, E., Collin, T., and Ehrlich, B. E. (2007) J. Neurosci. 27, 13813-13821). We now report the effect of O-GlcNAcylation on InsP(3)R-2 and InsP(3)R-3. Analysis of AR4-2J cells, a rat pancreatoma cell line expressing predominantly InsP(3)R-2, showed no detectable O-GlcNAcylation of InsP(3)R-2 and no significant functional changes despite the presence of the enzymes for addition (O-β-N-acetylglucosaminyltransferase) and removal (O-β-N-acetylglucosaminidase) of the monosaccharide. In contrast, InsP(3)R-3 in Mz-ChA-1 cells, a human cholangiocarcinoma cell line expressing predominantly InsP(3)R-3, was functionally modified by O-GlcNAcylation. Interestingly, the functional impact of O-GlcNAcylation on the InsP(3)R-3 channel was opposite the effect measured with InsP(3)R-1. Addition of O-GlcNAc by O-β-N-acetylglucosaminyltransferase increased InsP(3)R-3 single channel open probability. Incubation of Mz-ChA-1 cells in hyperglycemic medium caused an increase in the InsP(3)-dependent calcium release from the endoplasmic reticulum. The dynamic and inducible nature of O-GlcNAcylation and the InsP(3)R isoform specificity suggest that this form of modification of InsP(3)R and subsequent changes in intracellular calcium transients are important in physiological and pathophysiological processes.
Figures


Similar articles
-
Apoptosis regulation by Bcl-x(L) modulation of mammalian inositol 1,4,5-trisphosphate receptor channel isoform gating.Proc Natl Acad Sci U S A. 2007 Jul 24;104(30):12565-70. doi: 10.1073/pnas.0702489104. Epub 2007 Jul 16. Proc Natl Acad Sci U S A. 2007. PMID: 17636122 Free PMC article.
-
Regulation of the inositol 1,4,5-trisphosphate receptor type I by O-GlcNAc glycosylation.J Neurosci. 2007 Dec 12;27(50):13813-21. doi: 10.1523/JNEUROSCI.2069-07.2007. J Neurosci. 2007. PMID: 18077693 Free PMC article.
-
Apoptosis protection by Mcl-1 and Bcl-2 modulation of inositol 1,4,5-trisphosphate receptor-dependent Ca2+ signaling.J Biol Chem. 2010 Apr 30;285(18):13678-84. doi: 10.1074/jbc.M109.096040. Epub 2010 Feb 26. J Biol Chem. 2010. PMID: 20189983 Free PMC article.
-
Inositol 1,4,5-trisphosphate receptors in the endoplasmic reticulum: A single-channel point of view.Cell Calcium. 2015 Jul;58(1):67-78. doi: 10.1016/j.ceca.2014.12.008. Epub 2014 Dec 18. Cell Calcium. 2015. PMID: 25555684 Free PMC article. Review.
-
Inositol 1,4,5-trisphosphate receptor-isoform diversity in cell death and survival.Biochim Biophys Acta. 2014 Oct;1843(10):2164-83. doi: 10.1016/j.bbamcr.2014.03.007. Epub 2014 Mar 15. Biochim Biophys Acta. 2014. PMID: 24642269 Review.
Cited by
-
Type 3 Inositol 1,4,5-Trisphosphate Receptor Is Increased and Enhances Malignant Properties in Cholangiocarcinoma.Hepatology. 2020 Feb;71(2):583-599. doi: 10.1002/hep.30839. Epub 2019 Aug 19. Hepatology. 2020. PMID: 31251815 Free PMC article.
-
Nutrient Sensor mTOR and OGT: Orchestrators of Organelle Homeostasis in Pancreatic β-Cells.J Diabetes Res. 2020 Dec 16;2020:8872639. doi: 10.1155/2020/8872639. eCollection 2020. J Diabetes Res. 2020. PMID: 33457426 Free PMC article. Review.
-
Type 3 inositol 1,4,5-trisphosphate receptor: A calcium channel for all seasons.Cell Calcium. 2020 Jan;85:102132. doi: 10.1016/j.ceca.2019.102132. Epub 2019 Nov 25. Cell Calcium. 2020. PMID: 31790953 Free PMC article. Review.
-
Reduction in O-GlcNAcylation Mitigates the Severity of Inflammatory Response in Cerulein-Induced Acute Pancreatitis in a Mouse Model.Biology (Basel). 2022 Feb 22;11(3):347. doi: 10.3390/biology11030347. Biology (Basel). 2022. PMID: 35336721 Free PMC article.
-
Modulation of dynamin-related protein 1 (DRP1) function by increased O-linked-β-N-acetylglucosamine modification (O-GlcNAc) in cardiac myocytes.J Biol Chem. 2012 Aug 24;287(35):30024-34. doi: 10.1074/jbc.M112.390682. Epub 2012 Jun 28. J Biol Chem. 2012. PMID: 22745122 Free PMC article.
References
-
- Berridge M. J. (1993) Nature 361, 315–325 - PubMed
-
- Mikoshiba K. (2007) J. Neurochem. 102, 1426–1446 - PubMed
-
- Vermassen E., Parys J. B., Mauger J. P. (2004) Biol. Cell 96, 3–17 - PubMed
-
- Furuichi T., Mikoshiba K. (1995) J. Neurochem. 64, 953–960 - PubMed
-
- Patel S., Joseph S. K., Thomas A. P. (1999) Cell Calcium 25, 247–264 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

