Segregation of fluorescent membrane lipids into distinct micrometric domains: evidence for phase compartmentation of natural lipids?
- PMID: 21386970
- PMCID: PMC3046177
- DOI: 10.1371/journal.pone.0017021
Segregation of fluorescent membrane lipids into distinct micrometric domains: evidence for phase compartmentation of natural lipids?
Abstract
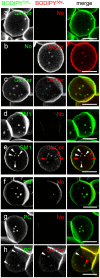
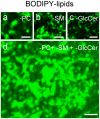
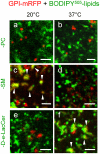
Background: We recently reported that sphingomyelin (SM) analogs substituted on the alkyl chain by various fluorophores (e.g. BODIPY) readily inserted at trace levels into the plasma membrane of living erythrocytes or CHO cells and spontaneously concentrated into micrometric domains. Despite sharing the same fluorescent ceramide backbone, BODIPY-SM domains segregated from similar domains labelled by BODIPY-D-e-lactosylceramide (D-e-LacCer) and depended on endogenous SM.
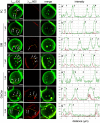
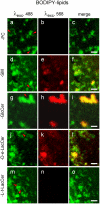
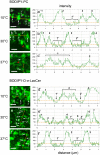
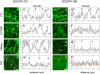
Methodology/principal findings: We show here that BODIPY-SM further differed from BODIPY-D-e-LacCer or -glucosylceramide (GlcCer) domains in temperature dependence, propensity to excimer formation, association with a glycosylphosphatidylinositol (GPI)-anchored fluorescent protein reporter, and lateral diffusion by FRAP, thus demonstrating different lipid phases and boundaries. Whereas BODIPY-D-e-LacCer behaved like BODIPY-GlcCer, its artificial stereoisomer, BODIPY-L-t-LacCer, behaved like BODIPY- and NBD-phosphatidylcholine (PC). Surprisingly, these two PC analogs also formed micrometric patches yet preferably at low temperature, did not show excimer, never associated with the GPI reporter and showed major restriction to lateral diffusion when photobleached in large fields. This functional comparison supported a three-phase micrometric compartmentation, of decreasing order: BODIPY-GSLs > -SM > -PC (or artificial L-t-LacCer). Co-existence of three segregated compartments was further supported by double labelling experiments and was confirmed by additive occupancy, up to ∼70% cell surface coverage. Specific alterations of BODIPY-analogs domains by manipulation of corresponding endogenous sphingolipids suggested that distinct fluorescent lipid partition might reflect differential intrinsic propensity of endogenous membrane lipids to form large assemblies.
Conclusions/significance: We conclude that fluorescent membrane lipids spontaneously concentrate into distinct micrometric assemblies. We hypothesize that these might reflect preexisting compartmentation of endogenous PM lipids into non-overlapping domains of differential order: GSLs > SM > PC, resulting into differential self-adhesion of the two former, with exclusion of the latter.
Conflict of interest statement
Figures


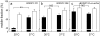
References
-
- Singer SJ, Nicolson GL. The fluid mosaic model of the structure of cell membranes. Science. 1972;175:720–731. - PubMed
-
- Singh RD, Liu Y, Wheatley CL, Holicky EL, Makino A, et al. Caveolar endocytosis and microdomain association of a glycosphingolipid analog is dependent on its sphingosine stereochemistry. J Biol Chem. 2006;281:30660–30668. - PubMed
-
- Brown DA, London E. Functions of lipid rafts in biological membranes. Annu Rev Cell Dev Biol. 1998;14:111–136. - PubMed
-
- Simons K, Ikonen E. Functional rafts in cell membranes. Nature. 1997;387:569–572. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

