Two phosphodiesterase genes, PDEL and PDEH, regulate development and pathogenicity by modulating intracellular cyclic AMP levels in Magnaporthe oryzae
- PMID: 21386978
- PMCID: PMC3046207
- DOI: 10.1371/journal.pone.0017241
Two phosphodiesterase genes, PDEL and PDEH, regulate development and pathogenicity by modulating intracellular cyclic AMP levels in Magnaporthe oryzae
Abstract
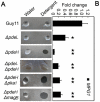
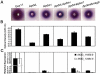
Cyclic AMP (cAMP) signaling plays an important role in regulating multiple cellular responses, such as growth, morphogenesis, and/or pathogenicity of eukaryotic organisms such as fungi. As a second messenger, cAMP is important in the activation of downstream effector molecules. The balance of intracellular cAMP levels depends on biosynthesis by adenylyl cyclases (ACs) and hydrolysis by cAMP phosphodiesterases (PDEases). The rice blast fungus Magnaporthe oryzae contains a high-affinity (PdeH/Pde2) and a low-affinity (PdeL/Pde1) PDEases, and a previous study showed that PdeH has a major role in asexual differentiation and pathogenicity. Here, we show that PdeL is required for asexual development and conidial morphology, and it also plays a minor role in regulating cAMP signaling. This is in contrast to PdeH whose mutation resulted in major defects in conidial morphology, cell wall integrity, and surface hydrophobicity, as well as a significant reduction in pathogenicity. Consistent with both PdeH and PdeL functioning in cAMP signaling, disruption of PDEH only partially rescued the mutant phenotype of ΔmagB and Δpka1. Further studies suggest that PdeH might function through a feedback mechanism to regulate the expression of pathogenicity factor Mpg1 during surface hydrophobicity and pathogenic development. Moreover, microarray data revealed new insights into the underlying cAMP regulatory mechanisms that may help to identify potential pathogenicity factors for the development of new disease management strategies.
Conflict of interest statement
Figures


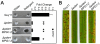
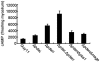


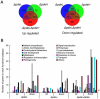
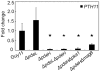
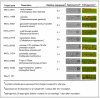
References
-
- Daniel PB, Walker WH, Habener JF. Cyclic AMP signaling and gene regulation. Annu Rev Nutr. 1998;18:353–83. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

