PACAP: a master regulator of neuroendocrine stress circuits and the cellular stress response
- PMID: 21388403
- PMCID: PMC3078626
- DOI: 10.1111/j.1749-6632.2011.05904.x
PACAP: a master regulator of neuroendocrine stress circuits and the cellular stress response
Abstract
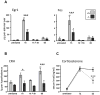
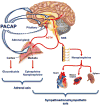
The neuropeptide pituitary adenylate cyclase-activating polypeptide (PACAP) is released from stress-transducing neurons. It exerts postsynaptic effects required to complete the hypothalamo-pituitary-adrenocortical (HPA) and hypothalamo-sympatho-adrenal (HSA) circuits activated by psychogenic and metabolic stressors. Upon activation of these circuits, PACAP-responsive (in cell culture models) and PACAP-dependent (in vivo) transcriptomic responses in the adrenal gland, hypothalamus, and pituitary have been identified. Gene products produced in response circuits during stress include additional neuropeptides, neurotransmitter biosynthetic enzymes, and neuroprotective factors. Major portions of HPA and HSA stress responses are abolished in PACAP-deficient mice. This deficit occurs at the level of both the hypothalamus (HPA axis) and the adrenal medulla (HSA axis). PACAP-dependent transcriptional stress responses are conveyed through noncanonical cyclic AMP- and calcium-initiated signaling pathways within the HSA circuit. PACAP transcriptional regulation of the HPA axis, in the hypothalamus, is likely to be mediated via canonical cyclic AMP signaling through protein kinase A.
© 2011 New York Academy of Sciences. No claim to original US Government works.
Figures
Similar articles
-
Pituitary adenylate cyclase-activating polypeptide controls stimulus-transcription coupling in the hypothalamic-pituitary-adrenal axis to mediate sustained hormone secretion during stress.J Neuroendocrinol. 2011 Oct;23(10):944-55. doi: 10.1111/j.1365-2826.2011.02202.x. J Neuroendocrinol. 2011. PMID: 21824204 Free PMC article.
-
Pituitary adenylate cyclase-activating polypeptide (PACAP): a master regulator in central and peripheral stress responses.Adv Pharmacol. 2013;68:445-57. doi: 10.1016/B978-0-12-411512-5.00021-X. Adv Pharmacol. 2013. PMID: 24054157 Review.
-
Stress hormone synthesis in mouse hypothalamus and adrenal gland triggered by restraint is dependent on pituitary adenylate cyclase-activating polypeptide signaling.Neuroscience. 2010 Feb 17;165(4):1025-30. doi: 10.1016/j.neuroscience.2009.11.023. Epub 2009 Nov 18. Neuroscience. 2010. PMID: 19931358 Free PMC article.
-
Perspectives on pituitary adenylate cyclase activating polypeptide (PACAP) in the neuroendocrine, endocrine, and nervous systems.Jpn J Physiol. 1998 Oct;48(5):301-31. doi: 10.2170/jjphysiol.48.301. Jpn J Physiol. 1998. PMID: 9852340 Review.
-
Role of VIP, PACAP, and related peptides in the regulation of the hypothalamo-pituitary-adrenal axis.Peptides. 1998;19(8):1443-67. doi: 10.1016/s0196-9781(98)00102-8. Peptides. 1998. PMID: 9809661 Review.
Cited by
-
Pituitary adenylate cyclase activating polypeptide (PACAP), stress, and sex hormones.Stress. 2017 Sep;20(5):465-475. doi: 10.1080/10253890.2017.1336535. Epub 2017 Jun 14. Stress. 2017. PMID: 28610473 Free PMC article. Review.
-
Neuropeptide regulation of signaling and behavior in the BNST.Mol Cells. 2015 Jan 31;38(1):1-13. doi: 10.14348/molcells.2015.2261. Epub 2014 Dec 4. Mol Cells. 2015. PMID: 25475545 Free PMC article. Review.
-
Circulating PACAP levels are associated with increased amygdala-default mode network resting-state connectivity in posttraumatic stress disorder.Neuropsychopharmacology. 2023 Jul;48(8):1245-1254. doi: 10.1038/s41386-023-01593-5. Epub 2023 May 9. Neuropsychopharmacology. 2023. PMID: 37161077 Free PMC article.
-
Circulating PACAP peptide and PAC1R genotype as possible transdiagnostic biomarkers for anxiety disorders in women: a preliminary study.Neuropsychopharmacology. 2020 Jun;45(7):1125-1133. doi: 10.1038/s41386-020-0604-4. Epub 2020 Jan 7. Neuropsychopharmacology. 2020. PMID: 31910434 Free PMC article.
-
The PACAP/PAC1 Receptor System and Feeding.Brain Sci. 2021 Dec 23;12(1):13. doi: 10.3390/brainsci12010013. Brain Sci. 2021. PMID: 35053757 Free PMC article. Review.
References
-
- Eiden LE. Signaling During Exocytosis. In: Bradshaw R, Dennis E, editors. Handbook of Cell Signaling. Vol. 3. Academic Press; New York: 2003. pp. 375–392.
-
- Hökfelt T, Bartfai T, Bloom F. Neuropeptides:opportunities for drug discovery. Lancet Neurology. 2003;2:463–472. - PubMed
-
- Miyata A, et al. Isolation of a novel 38 residue-hypothalamic polypeptide which stimulates adenylate cyclase in pituitary cells. Biochem Biophys Res Commun. 1989;164:567–574. - PubMed
-
- Mustafa T, Eiden LE. The Secretin Superfamily: PACAP, VIP and Related Peptides. In: Lim R, editor. Handbook of Neurochemistry and Molecular Neurobiology: XIII. Neuroactive Peptides and Proteins. XIII. Springer Heidelberg; 2006. pp. 1–36.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

