Using a business model approach and marketing techniques for recruitment to clinical trials
- PMID: 21396088
- PMCID: PMC3063210
- DOI: 10.1186/1745-6215-12-74
Using a business model approach and marketing techniques for recruitment to clinical trials
Abstract
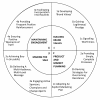
Randomised controlled trials (RCTs) are generally regarded as the gold standard for evaluating health care interventions. The level of uncertainty around a trial's estimate of effect is, however, frequently linked to how successful the trial has been in recruiting and retaining participants. As recruitment is often slower or more difficult than expected, with many trials failing to reach their target sample size within the timescale and funding originally envisaged, the results are often less reliable than they could have been. The high number of trials that require an extension to the recruitment period in order to reach the required sample size potentially delays the introduction of more effective therapies into routine clinical practice. Moreover, it may result in less research being undertaken as resources are redirected to extending existing trials rather than funding additional studies.Poor recruitment to publicly-funded RCTs has been much debated but there remains remarkably little clear evidence as to why many trials fail to recruit well, which recruitment methods work, in which populations and settings and for what type of intervention. One proposed solution to improving recruitment and retention is to adopt methodology from the business world to inform and structure trial management techniques.We review what is known about interventions to improve recruitment to trials. We describe a proposed business approach to trials and discuss the implementation of using a business model, using insights gained from three case studies.
Figures
References
-
- Pocock S. Clinical trials: a practical approach. Chichester: Wiley; 1983.
-
- Bowling A. Research methods in health: investigating health and health services. 2. Buckingham, Philadelphia: Open University Press; 2002.
-
- Prescott RJ, Counsell CE, Gillespie WJ, Grant AM, Russell IT, Kiauka S. et al.Factors that limit the number, quality and progress of randomised controlled trials. Health Technol Assess. 1999;3(20) - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials