Glucocorticoid regulation of human pulmonary surfactant protein-B (SP-B) mRNA stability is independent of activated glucocorticoid receptor
- PMID: 21398497
- PMCID: PMC3119123
- DOI: 10.1152/ajplung.00420.2010
Glucocorticoid regulation of human pulmonary surfactant protein-B (SP-B) mRNA stability is independent of activated glucocorticoid receptor
Abstract
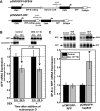
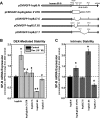
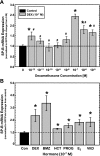
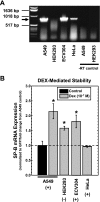
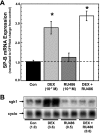
Adequate expression of surfactant protein-B (SP-B) is critical in the function of pulmonary surfactant to reduce alveolar surface tension. Expression of SP-B mRNA is restricted to specific lung-airway epithelial cells, and human SP-B mRNA stability is increased in the presence of the synthetic glucocorticoid dexamethasone (DEX). Although the mechanism of SP-B mRNA stabilization by DEX is unknown, studies suggest involvement of the glucocorticoid receptor (GR). We developed a dual-cistronic plasmid-based expression assay in which steady-state levels of SP-B mRNA, determined by Northern analysis, reproducibly reflect changes in SP-B mRNA stability. Using this assay, we found that steady-state levels of SP-B mRNA increased greater than twofold in transfected human-airway epithelial cells (A549) incubated with DEX (10(-7) M). DEX-mediated changes in SP-B mRNA levels required the presence of the SP-B mRNA 3'-untranslated region but did not require ongoing protein synthesis. The effect of DEX on SP-B mRNA levels was dose dependent, with maximal effect at 10(-7) M. DEX increased levels of SP-B mRNA in cells lacking GR, and the presence of the GR antagonist RU486 did not interfere with the effect of DEX. Surprisingly, other steroid hormones (progesterone, estradiol, and vitamin D; 10(-7) M) significantly increased SP-B mRNA levels, suggesting a common pathway of steroid hormone action on SP-B mRNA stability. These results indicate that the effect of DEX to increase SP-B mRNA stability is independent of activated GR and suggests that the mechanism is mediated by posttranscriptional or nongenomic effects of glucocorticoids.
Figures

Similar articles
-
A primate-specific RNA-binding protein (RBMXL3) is involved in glucocorticoid regulation of human pulmonary surfactant protein B (SP-B) mRNA stability.Am J Physiol Lung Cell Mol Physiol. 2021 May 1;320(5):L942-L957. doi: 10.1152/ajplung.00022.2020. Epub 2021 Mar 10. Am J Physiol Lung Cell Mol Physiol. 2021. PMID: 33719563 Free PMC article.
-
Glucocorticoid regulation of human pulmonary surfactant protein-B mRNA stability involves the 3'-untranslated region.Am J Respir Cell Mol Biol. 2008 Apr;38(4):473-82. doi: 10.1165/rcmb.2007-0303oc. Epub 2007 Nov 15. Am J Respir Cell Mol Biol. 2008. PMID: 18006875 Free PMC article.
-
Sequences of a hairpin structure in the 3'-untranslated region mediate regulation of human pulmonary surfactant protein B mRNA stability.Am J Physiol Lung Cell Mol Physiol. 2012 May 15;302(10):L1107-17. doi: 10.1152/ajplung.00015.2012. Epub 2012 Feb 24. Am J Physiol Lung Cell Mol Physiol. 2012. PMID: 22367784 Free PMC article.
-
Posttranscriptional regulation of surfactant protein-A messenger RNA in human fetal lung in vitro by glucocorticoids.Mol Endocrinol. 1991 Mar;5(3):414-23. doi: 10.1210/mend-5-3-414. Mol Endocrinol. 1991. PMID: 1653895
-
Dexamethasone potentiates keratinocyte growth factor-stimulated SP-A and SP-B gene expression in alveolar epithelial cells.Pediatr Res. 2003 Feb;53(2):231-9. doi: 10.1203/01.PDR.0000047840.77635.AE. Pediatr Res. 2003. PMID: 12538780
Cited by
-
Vitamin A supplementation prevents the bronchopulmonary dysplasia in premature infants: A systematic review and meta-analysis.Medicine (Baltimore). 2021 Jan 22;100(3):e23101. doi: 10.1097/MD.0000000000023101. Medicine (Baltimore). 2021. PMID: 33545924 Free PMC article.
-
Research progress on pathophysiologic mechanisms, clinical treatment and predictive biomarkers in bronchopulmonary dysplasia: from the perspective of oxidative stress.Front Pediatr. 2025 Mar 27;12:1343870. doi: 10.3389/fped.2024.1343870. eCollection 2024. Front Pediatr. 2025. PMID: 40212666 Free PMC article. Review.
-
Advances in tryptophan hydroxylase-2 gene expression regulation: new insights into serotonin-stress interaction and clinical implications.Am J Med Genet B Neuropsychiatr Genet. 2012 Mar;159B(2):152-71. doi: 10.1002/ajmg.b.32023. Am J Med Genet B Neuropsychiatr Genet. 2012. PMID: 22241550 Free PMC article. Review.
-
The problems of vitamin d insufficiency in older people.Aging Dis. 2012 Aug;3(4):313-29. Epub 2012 Jun 6. Aging Dis. 2012. PMID: 23185713 Free PMC article.
-
MicroRNAs 29b and 181a down-regulate the expression of the norepinephrine transporter and glucocorticoid receptors in PC12 cells.J Neurochem. 2016 Oct;139(2):197-207. doi: 10.1111/jnc.13761. Epub 2016 Sep 22. J Neurochem. 2016. PMID: 27501468 Free PMC article.
References
-
- Avery ME, Mead J. Surface properties in relation to atelectasis and hyaline membrane disease. Am J Dis Child 97: 517–523, 1959 - PubMed
-
- Baliga BS, Pronczuk AW, Munro HN. Mechanism of cycloheximide inhibition of protein synthesis in a cell-free system prepared from rat liver. J Biol Chem 244: 4480–4489, 1969 - PubMed
-
- Ballard PL, Ertsey R, Gonzales LW, Gonzales J. Transcriptional regulation of human pulmonary surfactant proteins SP-B and SP-C by glucocorticoids. Am J Respir Cell Mol Biol 14: 599–607, 1996 - PubMed
-
- Boggaram V, Qing K, Mendelson CR. The major apoprotein of rabbit pulmonary surfactant. Elucidation of primary sequence and cyclic AMP and developmental regulation. J Biol Chem 263: 2939–2947, 1988 - PubMed
-
- Cadepond F, Ulmann A, Baulieu EE. RU486 (mifepristone): mechanisms of action and clinical uses. Annu Rev Med 48: 129–156, 1997 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

