Role of the mar-sox-rob regulon in regulating outer membrane porin expression
- PMID: 21398557
- PMCID: PMC3133058
- DOI: 10.1128/JB.01382-10
Role of the mar-sox-rob regulon in regulating outer membrane porin expression
Abstract
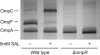
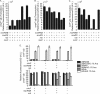
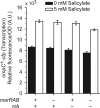
Multiple factors control the expression of the outer membrane porins OmpF and OmpC in Escherichia coli. In this work, we investigated the role of the mar-sox-rob regulon in regulating outer membrane porin expression in response to salicylate. We provide both genetic and physiological evidence that MarA and Rob can independently activate micF transcription in response to salicylate, leading to reduced OmpF expression. MarA was also found to repress OmpF expression through a MicF-independent pathway. In the case of OmpC, we found that its transcription was moderately increased in response to salicylate. However, this increase was independent of MarA and Rob. Finally, we found that the reduction in OmpF expression in a tolC mutant is due primarily to Rob. Collectively, this work further clarifies the coordinated role of MarA and Rob in regulating the expression of the outer membrane porins.
Figures


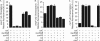

Similar articles
-
MarRA, SoxSR, and Rob encode a signal dependent regulatory network in Escherichia coli.Mol Biosyst. 2016 May 24;12(6):1901-12. doi: 10.1039/c6mb00263c. Mol Biosyst. 2016. PMID: 27098660
-
Regulation of ompF porin expression by salicylate in Escherichia coli.J Bacteriol. 1991 Sep;173(18):5631-8. doi: 10.1128/jb.173.18.5631-5638.1991. J Bacteriol. 1991. PMID: 1715858 Free PMC article.
-
Transcriptional cross talk within the mar-sox-rob regulon in Escherichia coli is limited to the rob and marRAB operons.J Bacteriol. 2012 Sep;194(18):4867-75. doi: 10.1128/JB.00680-12. Epub 2012 Jun 29. J Bacteriol. 2012. PMID: 22753060 Free PMC article.
-
MicF: an antisense RNA gene involved in response of Escherichia coli to global stress factors.J Mol Biol. 2001 Oct 12;313(1):1-12. doi: 10.1006/jmbi.2001.5029. J Mol Biol. 2001. PMID: 11601842 Review.
-
The Mar, Sox, and Rob Systems.EcoSal Plus. 2023 Dec 12;11(1):eesp00102022. doi: 10.1128/ecosalplus.esp-0010-2022. Epub 2023 Apr 4. EcoSal Plus. 2023. PMID: 37220096 Free PMC article. Review.
Cited by
-
Defining the function of OmpA in the Rcs stress response.Elife. 2020 Sep 28;9:e60861. doi: 10.7554/eLife.60861. Elife. 2020. PMID: 32985973 Free PMC article.
-
Multiscale regulation of nutrient stress responses in Escherichia coli from chromatin structure to small regulatory RNAs.bioRxiv [Preprint]. 2024 Jun 24:2024.06.20.599902. doi: 10.1101/2024.06.20.599902. bioRxiv. 2024. Update in: Nucleic Acids Res. 2025 Jul 8;53(13):gkaf647. doi: 10.1093/nar/gkaf647. PMID: 38979244 Free PMC article. Updated. Preprint.
-
Phenotypic and Multi-Omics Characterization of Escherichia coli K-12 Adapted to Chlorhexidine Identifies the Role of MlaA and Other Cell Envelope Alterations Regulated by Stress Inducible Pathways in CHX Resistance.Front Mol Biosci. 2021 May 19;8:659058. doi: 10.3389/fmolb.2021.659058. eCollection 2021. Front Mol Biosci. 2021. PMID: 34095221 Free PMC article.
-
Effects of Nucleobase Amino Acids on the Binding of Rob to Its Promoter DNA: Differential Alteration of DNA Affinity and Phenotype.Biochemistry. 2020 Jun 9;59(22):2111-2119. doi: 10.1021/acs.biochem.0c00290. Epub 2020 May 26. Biochemistry. 2020. PMID: 32412234 Free PMC article.
-
Antimicrobial Effects of Antipyretics.Antimicrob Agents Chemother. 2017 Mar 24;61(4):e02268-16. doi: 10.1128/AAC.02268-16. Print 2017 Apr. Antimicrob Agents Chemother. 2017. PMID: 28137805 Free PMC article. Review.
References
-
- Andersen J., Forst S. A., Zhao K., Inouye M., Delihas N. 1989. The function of micF RNA. micF RNA is a major factor in the thermal regulation of OmpF protein in Escherichia coli. J. Biol. Chem. 264:17961–17970 - PubMed
-
- Basle A., Rummel G., Storici P., Rosenbusch J. P., Schirmer T. 2006. Crystal structure of osmoporin OmpC from E. coli at 2.0 A. J. Mol. Biol. 362:933–942 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

