Next-Generation Antimalarial Drugs: Hybrid Molecules as a New Strategy in Drug Design
- PMID: 21399701
- PMCID: PMC3049227
- DOI: 10.1002/ddr.20345
Next-Generation Antimalarial Drugs: Hybrid Molecules as a New Strategy in Drug Design
Abstract
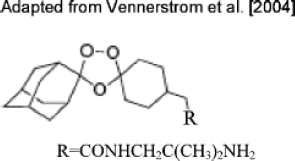
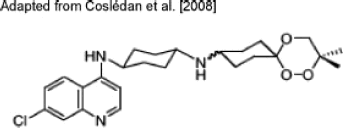
Malaria is a disease that affects nearly 40% of the global population, and chemotherapy remains the mainstay of its control strategy. The global malaria situation is increasingly being exacerbated by the emergence of drug resistance to most of the available antimalarials, necessitating search for novel drugs. A recent rational approach of antimalarial drug design characterized as "covalent bitherapy" involves linking two molecules with individual intrinsic activity into a single agent, thus packaging dual-activity into a single hybrid molecule. Current research in this field seems to endorse hybrid molecules as the next-generation antimalarial drugs. If the selective toxicity of hybrid prodrugs can be demonstrated in vivo with good bioavailability at the target site in the parasite, it would offer various advantages including dosage compliance, minimized toxicity, ability to design better drug combinations, and cheaper preclinical evaluation while achieving the ultimate object of delaying or circumventing the development of resistance. This review is focused on several hybrid molecules that have been developed, with particular emphasis on those deemed to have high potential for development for clinical use. Drug Dev Res 71: 20-32, 2010. © 2009 Wiley-Liss, Inc.
Figures

Similar articles
-
Novel rational drug design strategies with potential to revolutionize malaria chemotherapy.Curr Med Chem. 2011;18(1):113-43. doi: 10.2174/092986711793979742. Curr Med Chem. 2011. PMID: 21110811 Review.
-
Advancement of chimeric hybrid drugs to cure malaria infection: An overview with special emphasis on endoperoxide pharmacophores.Eur J Med Chem. 2021 Jul 5;219:113408. doi: 10.1016/j.ejmech.2021.113408. Epub 2021 Apr 4. Eur J Med Chem. 2021. PMID: 33989911 Review.
-
Chemical Synthesis, Efficacy, and Safety of Antimalarial Hybrid Drug Comprising of Sarcosine and Aniline Pharmacophores as Scaffolds.J Trop Med. 2020 Apr 9;2020:1643015. doi: 10.1155/2020/1643015. eCollection 2020. J Trop Med. 2020. PMID: 32328112 Free PMC article.
-
Considerations on the mechanism of action of artemisinin antimalarials: part 1--the 'carbon radical' and 'heme' hypotheses.Infect Disord Drug Targets. 2013 Aug;13(4):217-77. doi: 10.2174/1871526513666131129155708. Infect Disord Drug Targets. 2013. PMID: 24304352 Review.
-
Are Antimalarial Hybrid Molecules a Close Reality or a Distant Dream?Antimicrob Agents Chemother. 2017 Apr 24;61(5):e00249-17. doi: 10.1128/AAC.00249-17. Print 2017 May. Antimicrob Agents Chemother. 2017. PMID: 28289029 Free PMC article. Review.
Cited by
-
Synthesis of a potent antimalarial agent through natural products conjugation.ChemMedChem. 2013 Feb;8(2):221-5. doi: 10.1002/cmdc.201200503. Epub 2013 Jan 10. ChemMedChem. 2013. PMID: 23307699 Free PMC article.
-
Molecular docking and QSAR studies for modeling the antimalarial activity of hybrids 4-anilinoquinoline-triazines derivatives with the wild-type and mutant receptor pf-DHFR.Heliyon. 2019 Aug 26;5(8):e02357. doi: 10.1016/j.heliyon.2019.e02357. eCollection 2019 Aug. Heliyon. 2019. PMID: 31485537 Free PMC article.
-
Crystal structure of 9-amino-acridinium chloride N,N-di-methyl-formamide monosolvate.Acta Crystallogr E Crystallogr Commun. 2021 Nov 16;77(Pt 12):1303-1306. doi: 10.1107/S2056989021011816. eCollection 2021 Dec 1. Acta Crystallogr E Crystallogr Commun. 2021. PMID: 34925903 Free PMC article.
-
Indole: The After Next Scaffold of Antiplasmodial Agents?Drug Des Devel Ther. 2020 Nov 11;14:4855-4867. doi: 10.2147/DDDT.S278588. eCollection 2020. Drug Des Devel Ther. 2020. PMID: 33204071 Free PMC article. Review.
-
New Perspectives on How to Discover Drugs from Herbal Medicines: CAM's Outstanding Contribution to Modern Therapeutics.Evid Based Complement Alternat Med. 2013;2013:627375. doi: 10.1155/2013/627375. Epub 2013 Mar 24. Evid Based Complement Alternat Med. 2013. PMID: 23634172 Free PMC article.
References
-
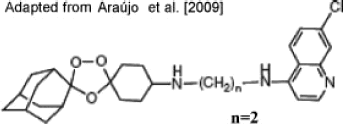
- Araújo NC, Barton V, Jones M, Stocks PA, Ward SA, Davies J, Bray PG, Shone AE, Cristiano ML, O'Neill PM. Semi-synthetic and synthetic 1,2,4-trioxaquines and 1,2,4-trioxolaquines: synthesis, preliminary SAR and comparison with acridine endoperoxide conjugates. Bioorg Med Chem Lett. 2009;19:2038–2043. - PubMed
-
- Atamna H, Ginsburg H. The malaria parasite supplies glutathione to its host cell: investigation of glutathione transport and metabolism in human erythrocytes infected with Plasmodium falciparum. Eur J Biochem. 1997;250:670–679. - PubMed
-
- Becker K, Rahlfs S, Nickel C, Schirmer RH. Glutathione: functions and metabolism in the malarial parasite Plasmodium falciparum. Biol Chem. 2003;384:551–566. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources

