X-ray microanalysis investigation of the changes in Na, K, and hemoglobin concentration in plasmodium falciparum-infected red blood cells
- PMID: 21402025
- PMCID: PMC3059598
- DOI: 10.1016/j.bpj.2011.02.007
X-ray microanalysis investigation of the changes in Na, K, and hemoglobin concentration in plasmodium falciparum-infected red blood cells
Abstract
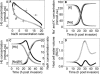
Plasmodium falciparum is responsible for severe malaria. During the ∼48 h duration of its asexual reproduction cycle in human red blood cells, the parasite causes profound alterations in the homeostasis of the host red cell, with reversal of the normal Na and K gradients across the host cell membrane, and a drastic fall in hemoglobin content. A question critical to our understanding of how the host cell retains its integrity for the duration of the cycle had been previously addressed by modeling the homeostasis of infected cells. The model predicted a critical contribution of excess hemoglobin consumption to cell integrity (the colloidosmotic hypothesis). Here we tested this prediction with the use of electron-probe x-ray microanalysis to measure the stage-related changes in Na, K, and Fe contents in single infected red cells and in uninfected controls. The results document a decrease in Fe signal with increased Na/K ratio. Interpreted in terms of concentrations, the results point to a sustained fall in host cell hemoglobin concentration with parasite maturation, supporting a colloidosmotic role of excess hemoglobin digestion. The results also provide, for the first time to our knowledge, comprehensive maps of the elemental distributions of Na, K, and Fe in falciparum-infected red blood cells.
Copyright © 2011 Biophysical Society. Published by Elsevier Inc. All rights reserved.
Figures
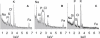


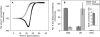

Similar articles
-
Biochemistry of malaria parasite infected red blood cells by X-ray microscopy.Sci Rep. 2017 Apr 11;7(1):802. doi: 10.1038/s41598-017-00921-2. Sci Rep. 2017. PMID: 28400621 Free PMC article.
-
Excess hemoglobin digestion and the osmotic stability of Plasmodium falciparum-infected red blood cells.Blood. 2003 May 15;101(10):4189-94. doi: 10.1182/blood-2002-08-2654. Epub 2003 Jan 16. Blood. 2003. PMID: 12531811
-
FRET imaging of hemoglobin concentration in Plasmodium falciparum-infected red cells.PLoS One. 2008;3(11):e3780. doi: 10.1371/journal.pone.0003780. Epub 2008 Nov 21. PLoS One. 2008. PMID: 19023444 Free PMC article.
-
Ion metabolism in malaria-infected erythrocytes.Blood Cells. 1990;16(2-3):437-49. Blood Cells. 1990. PMID: 2175223 Review.
-
Vesicle-mediated trafficking of parasite proteins to the host cell cytosol and erythrocyte surface membrane in Plasmodium falciparum infected erythrocytes.Int J Parasitol. 2001 Oct;31(12):1381-91. doi: 10.1016/s0020-7519(01)00256-9. Int J Parasitol. 2001. PMID: 11566305 Review.
Cited by
-
Malaria parasites tolerate a broad range of ionic environments and do not require host cation remodelling.Mol Microbiol. 2013 Apr;88(1):20-34. doi: 10.1111/mmi.12159. Epub 2013 Feb 26. Mol Microbiol. 2013. PMID: 23347042 Free PMC article.
-
Changes in K+ Concentration as a Signaling Mechanism in the Apicomplexa Parasites Plasmodium and Toxoplasma.Int J Mol Sci. 2023 Apr 14;24(8):7276. doi: 10.3390/ijms24087276. Int J Mol Sci. 2023. PMID: 37108438 Free PMC article. Review.
-
Soft X-ray microscopy analysis of cell volume and hemoglobin content in erythrocytes infected with asexual and sexual stages of Plasmodium falciparum.J Struct Biol. 2012 Feb;177(2):224-32. doi: 10.1016/j.jsb.2011.09.003. Epub 2011 Sep 16. J Struct Biol. 2012. PMID: 21945653 Free PMC article.
-
The malaria parasite cation ATPase PfATP4 and its role in the mechanism of action of a new arsenal of antimalarial drugs.Int J Parasitol Drugs Drug Resist. 2015 Aug 27;5(3):149-62. doi: 10.1016/j.ijpddr.2015.07.001. eCollection 2015 Dec. Int J Parasitol Drugs Drug Resist. 2015. PMID: 26401486 Free PMC article. Review.
-
Differential time-dependent volumetric and surface area changes and delayed induction of new permeation pathways in P. falciparum-infected hemoglobinopathic erythrocytes.Cell Microbiol. 2017 Feb;19(2):e12650. doi: 10.1111/cmi.12650. Epub 2016 Aug 25. Cell Microbiol. 2017. PMID: 27450804 Free PMC article.
References
-
- Kirk K. Membrane transport in the malaria-infected erythrocyte. Physiol. Rev. 2001;81:495–537. - PubMed
-
- Kanaani J., Ginsburg H. Compartment analysis of ATP in malaria-infected erythrocytes. Biochem. Int. 1988;17:451–459. - PubMed
-
- Kanaani J., Ginsburg H. Metabolic interconnection between the human malarial parasite Plasmodium falciparum and its host erythrocyte. Regulation of ATP levels by means of an adenylate translocator and adenylate kinase. J. Biol. Chem. 1989;264:3194–3199. - PubMed
-
- Ginsburg H. How and why does the malarial parasite permeabilize its host cell membrane? In: Benga G.H., Trager J.M., editors. Biomembranes: Basic & Medical Research. Springer-Verlag; Berlin, Germany: 1988. pp. 188–203.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

