Expression pattern of endothelin system components and localization of smooth muscle cells in the human pre-ovulatory follicle
- PMID: 21406445
- PMCID: PMC3079471
- DOI: 10.1093/humrep/der066
Expression pattern of endothelin system components and localization of smooth muscle cells in the human pre-ovulatory follicle
Abstract
Background: Whether ovarian follicular rupture involves contractile activity or not has been debated for decades. Recently, study in the rodents has indicated that an endogenously produced potent vasoconstrictive peptide, endothelin-2 (EDN2), may induce follicular constriction immediately prior to ovulation. This study was aimed to systematically characterize the human ovarian endothelin system and localize smooth muscle cells to assess the possible involvement of contractile activity in human ovulation.
Methods: This is a prospective experimental study. Study subjects were 20 women aged 20-38 years who underwent IVF owing to tubal or male factors. Expression patterns of messenger RNAs (mRNAs) for EDN1, EDN2, EDN3, endothelin-converting enzyme-1 (ECE1 and ECE2), endothelin receptor A (ET(A)) and ET(B) in the granulosa cells (GCs) and cumulus cells and endothelin peptide concentration in the pre-ovulatory follicles were measured at 36 h after hCG injection. In addition, localization of ovarian smooth muscle cells and endothelin receptor expression were determined in normal (non-IVF patient) ovaries.
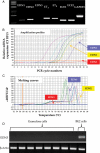
Results: Pre-ovulatory follicles express mRNA for EDN1 and EDN2, ECE1, ECE2, ET(A) and ET(B), but not EDN3, contain highly concentrated endothelin peptides (105.9 pg/ml) and are surrounded by theca externa that are made mostly of multicell layer non-vascular smooth muscle cells. ET(A) expression is localized in the smooth muscle cells of theca externa, theca interna and GC, whereas ET(B) expression is confined to theca interna.
Conclusions: Pre-ovulatory follicles contain highly concentrated endothelins and are surrounded by non-vascular smooth muscle cells that express endothelin receptor, suggesting involvement of endothelin-induced contractile action in ovulation in the human ovary.
Figures
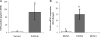



References
-
- Abberton KM, Healy DL, Rogers PA. Smooth muscle alpha actin and myosin heavy chain expression in the vascular smooth muscle cells surrounding human endometrial arterioles. Hum Reprod. 1999;14:3095–3100. - PubMed
-
- Al-Alem L, Bridges PJ, Su W, Gong MC, Iglarz M, Ko C. Endothelin-2 induces oviductal contraction via endothelin receptor subtype A in rats. J Endocrinol. 2007;193:383–391. - PubMed
-
- Amsterdam A, Lindner HR, Groschel-Stewart U. Localization of actin and myosin in the rat oocyte and follicular wall by immunofluorescence. Anat Rec. 1977;187:311–328. - PubMed
-
- Apa R, Miceli F, de Feo D, Pierro E, Ayala G, Mancuso S, Napolitano M, Lanzone A. Endothelin-1: expression and role in human corpus luteum. Am J Reprod Immunol. 1998;40:370–376. - PubMed
-
- Barton M, Yanagisawa M. Endothelin: 20 years from discovery to therapy. Can J Physiol Pharmacol. 2008;86:485–498. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

