Multifunctional transcription factor TFII-I is an activator of BRCA1 function
- PMID: 21407215
- PMCID: PMC3078593
- DOI: 10.1038/bjc.2011.75
Multifunctional transcription factor TFII-I is an activator of BRCA1 function
Abstract
Background: The TFII-I is a multifunctional transcriptional factor known to bind specifically to several DNA sequence elements and to mediate growth factor signalling. A microdeletion at the chromosomal location 7q11.23 encoding TFII-I and the related family of transcription factors may result in the onset of Williams-Beuren syndrome, an autosomal dominant genetic disorder characterised by a unique cognitive profile, diabetes, hypertension, anxiety, and craniofacial defects. Hereditary breast and ovarian cancer susceptibility gene product BRCA1 has been shown to serve as a positive regulator of SIRT1 expression by binding to the promoter region of SIRT1, but cross talk between BRCA1 and TFII-I has not been investigated to date.
Methods: A physical interaction between TFII-I and BRCA1 was explored. To determine pathophysiological function of TFII-I, its role as a transcriptional cofactor for BRCA1 was investigated.
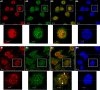
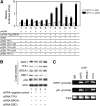
Results: We found a physical interaction between the carboxyl terminus of TFII-I and the carboxyl terminus of BRCA1, also known as the BRCT domain. Endogenous TFII-I and BRCA1 form a complex in nuclei of intact cells and formation of irradiation-induced nuclear foci was observed. We also showed that the expression of TFII-I stimulates the transcriptional activation function of BRCT by a transient expression assay. The expression of TFII-I also enhanced the transcriptional activation of the SIRT1 promoter mediated by full-length BRCA1.
Conclusion: These results revealed the intrinsic mechanism that TFII-I may modulate the cellular functions of BRCA1, and provide important implications to understand the development of breast cancer.
Figures


Similar articles
-
Identification of DBC1 as a transcriptional repressor for BRCA1.Br J Cancer. 2010 Mar 16;102(6):1061-7. doi: 10.1038/sj.bjc.6605577. Epub 2010 Feb 16. Br J Cancer. 2010. PMID: 20160719 Free PMC article.
-
Williams-Beuren syndrome-associated transcription factor TFII-I regulates osteogenic marker genes.J Biol Chem. 2009 Dec 25;284(52):36234-36239. doi: 10.1074/jbc.C109.063115. Epub 2009 Oct 30. J Biol Chem. 2009. PMID: 19880526 Free PMC article.
-
BRCA1 function mediates a TRAP/DRIP complex through direct interaction with TRAP220.Oncogene. 2004 Aug 5;23(35):6000-5. doi: 10.1038/sj.onc.1207786. Oncogene. 2004. Retraction in: Oncogene. 2014 Feb 6;33(6):804. doi: 10.1038/onc.2013.526. PMID: 15208681 Retracted.
-
Role of the multifunctional transcription factor TFII-I in DNA damage repair.DNA Repair (Amst). 2021 Oct;106:103175. doi: 10.1016/j.dnarep.2021.103175. Epub 2021 Jul 13. DNA Repair (Amst). 2021. PMID: 34280590 Review.
-
TFII-IDelta and TFII-Ibeta: unequal brothers fostering cellular proliferation.Mol Cell. 2006 Oct 20;24(2):169-71. doi: 10.1016/j.molcel.2006.10.003. Mol Cell. 2006. PMID: 17052451 Review.
Cited by
-
A novel crosstalk between BRCA1 and poly (ADP-ribose) polymerase 1 in breast cancer.Cell Cycle. 2014;13(21):3442-9. doi: 10.4161/15384101.2014.956507. Cell Cycle. 2014. PMID: 25485588 Free PMC article.
-
Identification of a peptide ligand for human ALDH3A1 through peptide phage display: Prediction and characterization of protein interaction sites and inhibition of ALDH3A1 enzymatic activity.Front Mol Biosci. 2023 Mar 20;10:1161111. doi: 10.3389/fmolb.2023.1161111. eCollection 2023. Front Mol Biosci. 2023. PMID: 37021113 Free PMC article.
-
The possible effects of the MTOR polymorphisms on preeclampsia susceptibility, severity, and onset: a case-control study and in silico analysis.Mol Biol Rep. 2024 Feb 23;51(1):335. doi: 10.1007/s11033-023-09190-x. Mol Biol Rep. 2024. PMID: 38393518
-
Role of multifunctional transcription factor TFII-I and putative tumour suppressor DBC1 in cell cycle and DNA double strand damage repair.Br J Cancer. 2013 Dec 10;109(12):3042-8. doi: 10.1038/bjc.2013.532. Epub 2013 Nov 14. Br J Cancer. 2013. PMID: 24231951 Free PMC article.
-
Anti-tumor activity of olaparib, a poly (ADP-ribose) polymerase (PARP) inhibitor, in cultured endometrial carcinoma cells.BMC Cancer. 2014 Mar 13;14:179. doi: 10.1186/1471-2407-14-179. BMC Cancer. 2014. PMID: 24625059 Free PMC article.
References
-
- Anderson SF, Schlegel BP, Nakajima T, Wolpin ES, Parvin JD (1998) BRCA1 protein is linked to the RNA polymerase II holoenzyme complex via RNA helicase A. Nat Genet 3: 254–256 - PubMed
-
- Bork P, Hofmann K, Bucher P, Neuwald AF, Altschul SF, Koonin EV (1997) A superfamily of conserved domains in DNA damage-responsive cell cycle checkpoint proteins. FASEB J 11: 68–76 - PubMed
-
- Callebaut I, Mornon JP (1997) From BRCA1 to RAP1: a widespread BRCT module closely associated with DNA repair. FEBS Lett 400: 25–30 - PubMed
-
- Chapman MS, Verma IM (1996) Transcriptional activation by BRCA1. Nature 382: 678–679 - PubMed
-
- Cheriyath V, Roy AL (2001) Structure-function analysis of TFII-I. Roles of the N-terminal end, basic region, and I-repeats. J Biol Chem 276: 8377–8383 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

