How can natural products serve as a viable source of lead compounds for the development of new/novel anti-malarials?
- PMID: 21411013
- PMCID: PMC3059460
- DOI: 10.1186/1475-2875-10-S1-S2
How can natural products serve as a viable source of lead compounds for the development of new/novel anti-malarials?
Abstract
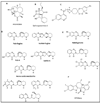
Malaria continues to be an enormous global health challenge, with millions of new infections and deaths reported annually. This is partly due to the development of resistance by the malaria parasite to the majority of established anti-malarial drugs, a situation that continues to hamper attempts at controlling the disease. This has spurred intensive drug discovery endeavours geared towards identifying novel, highly active anti-malarial drugs, and the identification of quality leads from natural sources would greatly augment these efforts. The current reality is that other than compounds that have their foundation in historic natural products, there are no other compounds in drug discovery as part of lead optimization projects and preclinical development or further that have originated from a natural product start-point in recent years. This paper briefly presents both classical as well as some more modern, but underutilized, approaches that have been applied outside the field of malaria, and which could be considered in enhancing the potential of natural products to provide or inspire the development of anti-malarial lead compounds.
Figures
Similar articles
-
Natural products as starting points for future anti-malarial therapies: going back to our roots?Malar J. 2011 Mar 15;10 Suppl 1(Suppl 1):S3. doi: 10.1186/1475-2875-10-S1-S3. Malar J. 2011. PMID: 21411014 Free PMC article. Review.
-
Drug resistance in Plasmodium: natural products in the fight against malaria.Mini Rev Med Chem. 2009 Feb;9(2):206-2124. doi: 10.2174/138955709787316074. Mini Rev Med Chem. 2009. PMID: 19200025 Review.
-
A call for using natural compounds in the development of new antimalarial treatments - an introduction.Malar J. 2011 Mar 15;10 Suppl 1(Suppl 1):S1. doi: 10.1186/1475-2875-10-S1-S1. Malar J. 2011. PMID: 21411010 Free PMC article. Review.
-
New innovations for an old infection: antimalarial lead discovery from marine natural products during the period 2003-2008.Future Med Chem. 2009 Jul;1(4):593-617. doi: 10.4155/fmc.09.56. Future Med Chem. 2009. PMID: 21426029 Review.
-
Comprehensive review on various strategies for antimalarial drug discovery.Eur J Med Chem. 2017 Jan 5;125:1300-1320. doi: 10.1016/j.ejmech.2016.11.025. Epub 2016 Nov 12. Eur J Med Chem. 2017. PMID: 27886547 Review.
Cited by
-
Antiplasmodial and Cytotoxic Activities of Toad Venoms from Southern Amazon, Brazil.Korean J Parasitol. 2016 Aug;54(4):415-21. doi: 10.3347/kjp.2016.54.4.415. Epub 2016 Aug 31. Korean J Parasitol. 2016. PMID: 27658592 Free PMC article.
-
CRIMALDDI: platform technologies and novel anti-malarial drug targets.Malar J. 2013 Nov 5;12:396. doi: 10.1186/1475-2875-12-396. Malar J. 2013. PMID: 24498961 Free PMC article.
-
Discovery and evaluation of thiazinoquinones as anti-protozoal agents.Mar Drugs. 2013 Sep 9;11(9):3472-99. doi: 10.3390/md11093472. Mar Drugs. 2013. PMID: 24022732 Free PMC article.
-
Alkaloids from Plants with Antimalarial Activity: A Review of Recent Studies.Evid Based Complement Alternat Med. 2020 Feb 12;2020:8749083. doi: 10.1155/2020/8749083. eCollection 2020. Evid Based Complement Alternat Med. 2020. PMID: 32104196 Free PMC article. Review.
-
Antiplasmodial activity of Xanthium strumarium against Plasmodium berghei-infected BALB/c mice.Parasitol Res. 2012 Mar;110(3):1179-83. doi: 10.1007/s00436-011-2611-1. Epub 2011 Aug 17. Parasitol Res. 2012. PMID: 21847597
References
-
- World Health Organization. World Malaria Report 2008
-
- World Health Organization. Guidelines for the treatment of malaria. 2006.
-
- World Health Organization Expert Committee on Malaria. WHO Technical Report Series 2000 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

