Histochemical localization of caldesmon in the CNS and ganglia of the mouse
- PMID: 21411712
- PMCID: PMC3201179
- DOI: 10.1369/0022155411400875
Histochemical localization of caldesmon in the CNS and ganglia of the mouse
Abstract
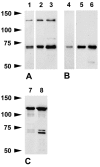
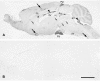
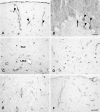
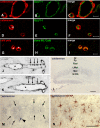
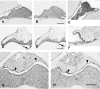
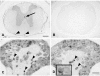
The author has recently reported the distribution of the cytoskeleton-associated protein caldesmon in spleen and lymph nodes detected with different antibodies against caldesmon (J Histochem Cytochem 58:183-193, 2010). Here the author reports the distribution of caldesmon in the CNS and ganglia of the mouse using the same antibodies. Western blot analysis of mouse brain and spinal cord showed the preponderance of l-caldesmon and suggested at least two l-caldesmon isoforms in the brain. Immunostaining revealed the predominant reactivity of smooth muscle cells and cells resembling pericytes of many large and small blood vessels, ependymocytes, and secretory cells of the pineal gland and pituitary gland. Neuronal perikarya and neuropil in general displayed no or weak immunoreactivity, but there was stronger labeling of neuronal perikarya in dorsal root and trigeminal ganglia. In the brain, staining of the neuropil was stronger in the molecular layers of the dentate gyrus and cerebellum. Results show that caldesmon is expressed in many different cell types in the CNS and ganglia, consistent with the notion that l-caldesmon is ubiquitously expressed, but it appears most concentrated in smooth muscle cells, pericytes, epithelial cells, secretory cells, and neuronal perikarya in dorsal root and trigeminal ganglia.
Conflict of interest statement
The author(s) declared no potential conflicts of interest with respect to the authorship and/or publication of this article.
Figures
Similar articles
-
Histochemical localization of caldesmon isoforms in colon adenocarcinoma and lymph node metastases.Virchows Arch. 2011 Jul;459(1):81-9. doi: 10.1007/s00428-011-1091-0. Epub 2011 May 29. Virchows Arch. 2011. PMID: 21626272
-
The actin-binding protein caldesmon is in spleen and lymph nodes predominately expressed by smooth-muscle cells, reticular cells, and follicular dendritic cells.J Histochem Cytochem. 2010 Feb;58(2):183-93. doi: 10.1369/jhc.2009.954651. J Histochem Cytochem. 2010. PMID: 19875849 Free PMC article.
-
Expression of the short chain fatty acid receptor GPR41/FFAR3 in autonomic and somatic sensory ganglia.Neuroscience. 2015 Apr 2;290:126-37. doi: 10.1016/j.neuroscience.2015.01.040. Epub 2015 Jan 28. Neuroscience. 2015. PMID: 25637492
-
Cytochemistry of the trigeminal and dorsal root ganglia and spinal cord of the rat.Comp Biochem Physiol A Comp Physiol. 1989;93(1):183-93. doi: 10.1016/0300-9629(89)90206-5. Comp Biochem Physiol A Comp Physiol. 1989. PMID: 2568221 Review.
-
Caldesmon.Curr Opin Cell Biol. 1993 Feb;5(1):70-6. doi: 10.1016/s0955-0674(05)80010-9. Curr Opin Cell Biol. 1993. PMID: 8448032 Review.
Cited by
-
Deep tissue analysis of distal aqueous drainage structures and contractile features.Sci Rep. 2017 Dec 6;7(1):17071. doi: 10.1038/s41598-017-16897-y. Sci Rep. 2017. PMID: 29213129 Free PMC article.
-
Host tissue response to floating microelectrode arrays chronically implanted in the feline spinal nerve.J Neural Eng. 2020 Jul 10;17(4):046012. doi: 10.1088/1741-2552/ab94d7. J Neural Eng. 2020. PMID: 32434161 Free PMC article.
-
Pericyte Antigens in Perivascular Soft Tissue Tumors.Int J Surg Pathol. 2015 Dec;23(8):638-48. doi: 10.1177/1066896915591272. Epub 2015 Jun 17. Int J Surg Pathol. 2015. PMID: 26085647 Free PMC article.
-
Histochemical localization of caldesmon isoforms in colon adenocarcinoma and lymph node metastases.Virchows Arch. 2011 Jul;459(1):81-9. doi: 10.1007/s00428-011-1091-0. Epub 2011 May 29. Virchows Arch. 2011. PMID: 21626272
-
Caldesmon: Biochemical and Clinical Implications in Cancer.Front Cell Dev Biol. 2021 Feb 18;9:634759. doi: 10.3389/fcell.2021.634759. eCollection 2021. Front Cell Dev Biol. 2021. PMID: 33681215 Free PMC article. Review.
References
-
- Abd-el-Basset EM, Ahmed I, Fedoroff S. 1991. Actin and actin-binding proteins in differentiating astroglia in tissue culture. J Neurosci Res. 30:1–17 - PubMed
-
- Agassandian C, Plantier M, Fattoum A, Represa A, der Terrossian E. 2000. Subcellular distribution of calponin and caldesmon in rat hippocampus. Brain Res. 887:444–449 - PubMed
-
- Allt G, Lawrenson JG. 2001. Pericytes: cell biology and pathology. Cells Tissues Organs. 169:1–11 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

