A postsleep decline in auditory evoked potential amplitude reflects sleep homeostasis
- PMID: 21420904
- PMCID: PMC3134628
- DOI: 10.1016/j.clinph.2011.01.041
A postsleep decline in auditory evoked potential amplitude reflects sleep homeostasis
Abstract
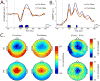
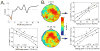
Objective: It has been hypothesized that slow wave activity, a well established measure of sleep homeostasis that increases after waking and decreases after sleep, may reflect changes in cortical synaptic strength. If so, the amplitude of sensory evoked responses should also vary as a function of time awake and asleep in a way that reflects sleep homeostasis.
Methods: Using 256-channel, high-density electroencephalography (EEG) in 12 subjects, auditory evoked potentials (AEP) and spontaneous waking data were collected during wakefulness before and after sleep.
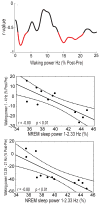
Results: The amplitudes of the N1 and P2 waves of the AEP were reduced after a night of sleep. In addition, the decline in N1 amplitude correlated with low-frequency EEG power during non-rapid eye movement sleep and spontaneous wakefulness, both homeostatically regulated measures of sleep need.
Conclusions: The decline in AEP amplitude after a night of sleep may reflect a homeostatic reduction in synaptic strength.
Significance: These findings provide further evidence for a connection between synaptic plasticity and sleep homeostasis.
Copyright © 2011 International Federation of Clinical Neurophysiology. Published by Elsevier Ireland Ltd. All rights reserved.
Conflict of interest statement
The other authors have indicated no financial conflicts of interest.
Figures
References
-
- Aeschbach D, Matthews JR, Postolache TT, Jackson MA, Giesen HA, Wehr TA. Two circadian rhythms in the human electroencephalogram during wakefulness. Am J Physiol. 1999;227(6 Pt 2):R1771–9. - PubMed
-
- Amzica F, Steriade M. Electrophysiological correlates of sleep delta waves. Electroenceph Clin Neurophysiol. 1998;107(2):69–83. - PubMed
-
- Baumann S, Meyer M, Jäncke L. Enhancement of Auditory-evoked Potentials in Musicians Reflects an Influence of Expertise but not Selective Attention. J Cogn Neurosci. 2008;20(12):2238–49. - PubMed
-
- Borbély AA. A Two Process Model of Sleep Regulation. Hum Neurobiol. 1982;1(3):195–204. - PubMed
-
- Cajochen C, Wyatt JK, Czeisler CA, Dijk DJ. Separation of Circadian and Wake Duration-Dependent Modulation of EEG Activation During Wakefulness. Neuroscience. 2002;114(4):1047–60. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

