Decrease in penicillin susceptibility due to heat shock protein ClpL in Streptococcus pneumoniae
- PMID: 21422206
- PMCID: PMC3101445
- DOI: 10.1128/AAC.01383-10
Decrease in penicillin susceptibility due to heat shock protein ClpL in Streptococcus pneumoniae
Abstract
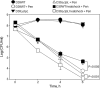
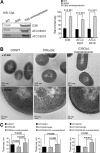
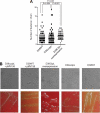
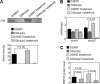
Antibiotic resistance and tolerance are increasing threats to global health as antibiotic-resistant bacteria can cause severe morbidity and mortality and can increase treatment cost 10-fold. Although several genes contributing to antibiotic tolerance among pneumococci have been identified, we report here that ClpL, a major heat shock protein, could modulate cell wall biosynthetic enzymes and lead to decreased penicillin susceptibility. On capsular type 1, 2, and 19 genetic backgrounds, mutants lacking ClpL were more susceptible to penicillin and had thinner cell walls than the parental strains, whereas a ClpL-overexpressing strain showed a higher resistance to penicillin and a thicker cell wall. Although exposure of Streptococcus pneumoniae D39 to penicillin inhibited expression of the major cell wall synthesis gene pbp2x, heat shock induced a ClpL-dependent increase in the mRNA levels and protein synthesized by pbp2x. Inducible ClpL expression correlated with PBP2x expression and penicillin susceptibility. Fractionation and electron micrograph data revealed that ClpL induced by heat shock is localized at the cell wall, and the ΔclpL showed significantly reduced net translocation of PBP2x into the cell wall. Moreover, coimmunoprecipitation with either ClpL or PBP2x antibody followed by reprobing with ClpL or PBP2x antibody showed an interaction between ClpL and PBP2x after heat stress. This interaction was confirmed by His tag pulldown assay with either ClpLHis₆ or PBP2xHis₆. Thus, ClpL stabilized pbp2x expression, interacted with PBP2x, and facilitated translocation of PBP2x, a key protein of cell wall synthesis process, contributing to the decrease of antibiotic susceptibility in S. pneumoniae.
Figures



Similar articles
-
Heat-shock protein ClpL/HSP100 increases penicillin tolerance in Streptococcus pneumoniae.Adv Otorhinolaryngol. 2011;72:126-8. doi: 10.1159/000324658. Epub 2011 Aug 18. Adv Otorhinolaryngol. 2011. PMID: 21865709
-
[Evaluation of penicillin-binding protein genotypes in penicillin susceptible and resistant Streptococcus pneumoniae isolates].Mikrobiyol Bul. 2012 Apr;46(2):190-201. Mikrobiyol Bul. 2012. PMID: 22639308 Turkish.
-
Diversity of Mosaic pbp2x Families in Penicillin-Resistant Streptococcus pneumoniae from Iran and Romania.Antimicrob Agents Chemother. 2017 Nov 22;61(12):e01535-17. doi: 10.1128/AAC.01535-17. Print 2017 Dec. Antimicrob Agents Chemother. 2017. PMID: 28971878 Free PMC article.
-
Alterations of the penicillin-binding proteins and murM alleles of clinical Streptococcus pneumoniae isolates with high-level resistance to amoxicillin in Spain.J Antimicrob Chemother. 2006 Feb;57(2):224-9. doi: 10.1093/jac/dki442. Epub 2005 Dec 20. J Antimicrob Chemother. 2006. PMID: 16368701
-
Phenotypes and genes of resistance of pneumococci to penicillin isolated from children.Prilozi. 2009 Jul;30(1):143-54. Prilozi. 2009. PMID: 19736537
Cited by
-
The physiology and genetics of bacterial responses to antibiotic combinations.Nat Rev Microbiol. 2022 Aug;20(8):478-490. doi: 10.1038/s41579-022-00700-5. Epub 2022 Mar 3. Nat Rev Microbiol. 2022. PMID: 35241807 Review.
-
Screening and identification of ClpE interaction proteins in Streptococcus pneumoniae by a bacterial two-hybrid system and co-immunoprecipitation.J Microbiol. 2013 Aug;51(4):453-60. doi: 10.1007/s12275-013-3001-4. Epub 2013 Aug 30. J Microbiol. 2013. PMID: 23990296
-
Stress responses in Streptococcus species and their effects on the host.J Microbiol. 2015 Nov;53(11):741-9. doi: 10.1007/s12275-015-5432-6. Epub 2015 Oct 28. J Microbiol. 2015. PMID: 26502957 Review.
-
Discovery of β-lactam-resistant variants in diverse pneumococcal populations.Genome Med. 2014 Sep 25;6(9):72. doi: 10.1186/s13073-014-0072-8. eCollection 2014. Genome Med. 2014. PMID: 25473434 Free PMC article.
-
Adenylate kinase from Streptococcus pneumoniae is essential for growth through its catalytic activity.FEBS Open Bio. 2014 Jul 8;4:672-82. doi: 10.1016/j.fob.2014.07.002. eCollection 2014. FEBS Open Bio. 2014. PMID: 25180151 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

