Molecular and functional characteristics of ovarian surface epithelial cells transformed by KrasG12D and loss of Pten in a mouse model in vivo
- PMID: 21423204
- PMCID: PMC3139785
- DOI: 10.1038/onc.2011.70
Molecular and functional characteristics of ovarian surface epithelial cells transformed by KrasG12D and loss of Pten in a mouse model in vivo
Abstract
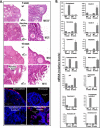
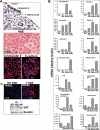
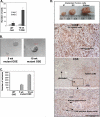
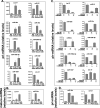
Ovarian cancer is a complex and deadly disease that remains difficult to detect at an early curable stage. Furthermore, although some oncogenic (Kras, Pten/PI3K and Trp53) pathways that are frequently mutated, deleted or amplified in ovarian cancer are known, how these pathways initiate and drive specific morphological phenotypes and tumor outcomes remain unclear. We recently generated Pten(fl/fl); Kras(G12D); Amhr2-Cre mice to disrupt the Pten gene and express a stable mutant form of Kras(G12D) in ovarian surface epithelial (OSE) cells. On the basis of histopathologic criteria, the mutant mice developed low-grade ovarian serous papillary adenocarcinomas at an early age and with 100% penetrance. This highly reproducible phenotype provides the first mouse model in which to study this ovarian cancer subtype. OSE cells isolated from ovaries of mutant mice at 5 and 10 weeks of age exhibit temporal changes in the expression of specific Mullerian epithelial marker genes, grow in soft agar and develop ectopic invasive tumors in recipient mice, indicating that the cells are transformed. Gene profiling identified specific mRNAs and microRNAs differentially expressed in purified OSE cells derived from tumors of the mutant mice compared with wild-type OSE cells. Mapping of transcripts or genes between the mouse OSE mutant data sets, the Kras signature from human cancer cell lines and the human ovarian tumor array data sets, documented significant overlap, indicating that KRAS is a key driver of OSE transformation in this context. Two key hallmarks of the mutant OSE cells in these mice are the elevated expression of the tumor-suppressor Trp53 (p53) and its microRNA target, miR-34a-c. We propose that elevated TRP53 and miR-34a-c may exert negatively regulatory effects that reduce the proliferative potential of OSE cells leading to the low-grade serous adenocarcinoma phenotype.
Figures



Similar articles
-
Wild-type tumor repressor protein 53 (Trp53) promotes ovarian cancer cell survival.Endocrinology. 2012 Apr;153(4):1638-48. doi: 10.1210/en.2011-2131. Epub 2012 Mar 6. Endocrinology. 2012. PMID: 22396451 Free PMC article.
-
Cell type-specific targeted mutations of Kras and Pten document proliferation arrest in granulosa cells versus oncogenic insult to ovarian surface epithelial cells.Cancer Res. 2009 Aug 15;69(16):6463-72. doi: 10.1158/0008-5472.CAN-08-3363. Cancer Res. 2009. PMID: 19679546 Free PMC article.
-
Modelling genetic and clinical heterogeneity in epithelial ovarian cancers.Carcinogenesis. 2011 Oct;32(10):1540-9. doi: 10.1093/carcin/bgr140. Epub 2011 Aug 22. Carcinogenesis. 2011. PMID: 21859834
-
Low Grade Serous Ovarian Carcinoma: from the molecular characterization to the best therapeutic strategy.Cancer Treat Rev. 2015 Feb;41(2):136-43. doi: 10.1016/j.ctrv.2014.12.003. Epub 2014 Dec 23. Cancer Treat Rev. 2015. PMID: 25573350 Review.
-
[Advances in origin and pathogenesis of epithelial ovarian cancer].Zhonghua Bing Li Xue Za Zhi. 2011 Aug;40(8):569-72. Zhonghua Bing Li Xue Za Zhi. 2011. PMID: 22169654 Review. Chinese. No abstract available.
Cited by
-
Identification of ovarian high-grade serous carcinoma cell lines that show estrogen-sensitive growth as xenografts in immunocompromised mice.Sci Rep. 2020 Jul 1;10(1):10799. doi: 10.1038/s41598-020-67533-1. Sci Rep. 2020. PMID: 32612269 Free PMC article.
-
Evaluating the progenitor cells of ovarian cancer: analysis of current animal models.BMB Rep. 2011 Jul;44(7):435-45. doi: 10.5483/BMBRep.2011.44.7.435. BMB Rep. 2011. PMID: 21777513 Free PMC article. Review.
-
Ubiquitin E3 ligase CRL4(CDT2/DCAF2) as a potential chemotherapeutic target for ovarian surface epithelial cancer.J Biol Chem. 2013 Oct 11;288(41):29680-91. doi: 10.1074/jbc.M113.495069. Epub 2013 Aug 30. J Biol Chem. 2013. PMID: 23995842 Free PMC article.
-
Dysregulation of mitotic machinery genes precedes genome instability during spontaneous pre-malignant transformation of mouse ovarian surface epithelial cells.BMC Genomics. 2016 Oct 25;17(Suppl 8):728. doi: 10.1186/s12864-016-3068-5. BMC Genomics. 2016. PMID: 27801298 Free PMC article.
-
Immunobiology of human mucin 1 in a preclinical ovarian tumor model.Oncogene. 2013 Aug 8;32(32):3664-75. doi: 10.1038/onc.2012.397. Epub 2012 Sep 10. Oncogene. 2013. PMID: 22964632 Free PMC article.
References
-
- Abdullah M, Schultz H, K. D, Branscheid D, Dalhoff K, Zabel P, et al. Expression of the acute phase protein haptoglobin in human lung cancer and tumor-free lung tissues. Pathol Res Pract. 2009;205:639–647. - PubMed
-
- Auersperg N, Wong AST, Choi K-C, Kang SK, Leung PCK. Ovarian surface epithelium: biology, endocrinology and pathology. Endocrine Rev. 2001;22:255–288. - PubMed
-
- Auersperg N, Woo MMM, Gilks CB. The origin of ovarian carcinomas: a developmental view. Gynecol Oncol. 2008;110:452–454. - PubMed
-
- Bohlig L, Metzger R, Rother K, Till H, Engeland K. The CCN3 gene coding an extracellular adhesion-related protein is transcriptionaly activated by the p53 tumor suppressor. Cell Cycle. 2008;7:1254–1261. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

