Ammonia production, excretion, toxicity, and defense in fish: a review
- PMID: 21423375
- PMCID: PMC3059970
- DOI: 10.3389/fphys.2010.00134
Ammonia production, excretion, toxicity, and defense in fish: a review
Abstract
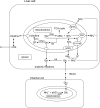
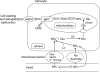
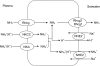
Many fishes are ammonotelic but some species can detoxify ammonia to glutamine or urea. Certain fish species can accumulate high levels of ammonia in the brain or defense against ammonia toxicity by enhancing the effectiveness of ammonia excretion through active NH4+transport, manipulation of ambient pH, or reduction in ammonia permeability through the branchial and cutaneous epithelia. Recent reports on ammonia toxicity in mammalian brain reveal the importance of permeation of ammonia through the blood-brain barrier and passages of ammonia and water through transporters in the plasmalemma of brain cells. Additionally, brain ammonia toxicity could be related to the passage of glutamine through the mitochondrial membranes into the mitochondrial matrix. On the other hand, recent reports on ammonia excretion in fish confirm the involvement of Rhesus glycoproteins in the branchial and cutaneous epithelia. Therefore, this review focuses on both the earlier literature and the up-to-date information on the problems and mechanisms concerning the permeation of ammonia, as NH(3), NH4+ or proton-neutral nitrogenous compounds, across mitochondrial membranes, the blood-brain barrier, the plasmalemma of neurons, and the branchial and cutaneous epithelia of fish. It also addresses how certain fishes with high ammonia tolerance defend against ammonia toxicity through the regulation of the permeation of ammonia and related nitrogenous compounds through various types of membranes. It is hoped that this review would revive the interests in investigations on the passage of ammonia through the mitochondrial membranes and the blood-brain barrier of ammonotelic fishes and fishes with high brain ammonia tolerance, respectively.
Keywords: ammonia; ammonia excretion; ammonia toxicity; ammonia transporter; fish; nitrogen metabolism.
Figures


References
-
- Anderson P. M. (2001). “Urea and glutamine synthesis: environmental influences on nitrogen excretion,” in Fish Physiology, Vol. 20, Nitrogen Excretion, edsWright P. A., Anderson P. M.New York: Academic Press; ), 239–277
-
- Anderson P. M., Broderius M. A., Fong K. C., Tsui T. K. N., Chew S. F., Ip Y. K. (2002). Glutamine synthetase expression in liver, muscle, stomach and intestine of Bostrichyths sinensis in response to exposure to a high exogenous ammonia concentration. J. Exp. Biol. 205, 2053–2065 - PubMed
-
- Anderson P. M., Casey C. A. (1984). Glutamine-dependent synthesis of citrulline by isolated hepatic mitochondria from Squalus acanthias. J. Biol. Chem. 259, 456–462 - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources

