Epithelial cell stretching and luminal acidification lead to a retarded development of stria vascularis and deafness in mice lacking pendrin
- PMID: 21423764
- PMCID: PMC3056798
- DOI: 10.1371/journal.pone.0017949
Epithelial cell stretching and luminal acidification lead to a retarded development of stria vascularis and deafness in mice lacking pendrin
Abstract
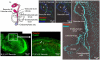
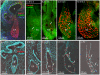
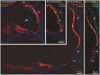
Loss-of-function mutations of SLC26A4/pendrin are among the most prevalent causes of deafness. Deafness and vestibular dysfunction in the corresponding mouse model, Slc26a4(-/-), are associated with an enlargement and acidification of the membranous labyrinth. Here we relate the onset of expression of the HCO(3) (-) transporter pendrin to the luminal pH and to enlargement-associated epithelial cell stretching. We determined expression with immunocytochemistry, cell stretching by digital morphometry and pH with double-barreled ion-selective electrodes. Pendrin was first expressed in the endolymphatic sac at embryonic day (E) 11.5, in the cochlear hook-region at E13.5, in the utricle and saccule at E14.5, in ampullae at E16.5, and in the upper turn of the cochlea at E17.5. Epithelial cell stretching in Slc26a4(-/-) mice began at E14.5. pH changes occurred first in the cochlea at E15.5 and in the endolymphatic sac at E17.5. At postnatal day 2, stria vascularis, outer sulcus and Reissner's membrane epithelial cells, and utricular and saccular transitional cells were stretched, whereas sensory cells in the cochlea, utricle and saccule did not differ between Slc26a4(+/-) and Slc26a4(-/-) mice. Structural development of stria vascularis, including vascularization, was retarded in Slc26a4(-/-) mice. In conclusion, the data demonstrate that the enlargement and stretching of non-sensory epithelial cells precedes luminal acidification in the cochlea and the endolymphatic sac. Stretching and luminal acidification may alter cell-to-cell communication and lead to the observed retarded development of stria vascularis, which may be an important step on the path to deafness in Slc26a4(-/-) mice, and possibly in humans, lacking functional pendrin expression.
Conflict of interest statement
Figures


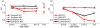




Similar articles
-
Loss of cochlear HCO3- secretion causes deafness via endolymphatic acidification and inhibition of Ca2+ reabsorption in a Pendred syndrome mouse model.Am J Physiol Renal Physiol. 2007 May;292(5):F1345-53. doi: 10.1152/ajprenal.00487.2006. Epub 2007 Feb 13. Am J Physiol Renal Physiol. 2007. PMID: 17299139 Free PMC article.
-
Free radical stress-mediated loss of Kcnj10 protein expression in stria vascularis contributes to deafness in Pendred syndrome mouse model.Am J Physiol Renal Physiol. 2008 Jan;294(1):F139-48. doi: 10.1152/ajprenal.00433.2007. Epub 2007 Oct 24. Am J Physiol Renal Physiol. 2008. PMID: 17959752 Free PMC article.
-
Failure of fluid absorption in the endolymphatic sac initiates cochlear enlargement that leads to deafness in mice lacking pendrin expression.PLoS One. 2010 Nov 17;5(11):e14041. doi: 10.1371/journal.pone.0014041. PLoS One. 2010. PMID: 21103348 Free PMC article.
-
Mouse models for pendrin-associated loss of cochlear and vestibular function.Cell Physiol Biochem. 2013;32(7):157-65. doi: 10.1159/000356635. Epub 2013 Dec 18. Cell Physiol Biochem. 2013. PMID: 24429822 Free PMC article. Review.
-
Potassium ion recycling pathway via gap junction systems in the mammalian cochlea and its interruption in hereditary nonsyndromic deafness.Med Electron Microsc. 2000;33(2):51-6. doi: 10.1007/s007950070001. Med Electron Microsc. 2000. PMID: 11810458 Review.
Cited by
-
Genetic Determinants of Non-Syndromic Enlarged Vestibular Aqueduct: A Review.Audiol Res. 2021 Aug 28;11(3):423-442. doi: 10.3390/audiolres11030040. Audiol Res. 2021. PMID: 34562878 Free PMC article. Review.
-
Progressive irreversible hearing loss is caused by stria vascularis degeneration in an Slc26a4-insufficient mouse model of large vestibular aqueduct syndrome.Neuroscience. 2015 Dec 3;310:188-97. doi: 10.1016/j.neuroscience.2015.09.016. Epub 2015 Sep 9. Neuroscience. 2015. PMID: 26363152 Free PMC article.
-
Molecular architecture underlying fluid absorption by the developing inner ear.Elife. 2017 Oct 10;6:e26851. doi: 10.7554/eLife.26851. Elife. 2017. PMID: 28994389 Free PMC article.
-
Lmx1a is essential for marginal cell differentiation and stria vascularis formation.Front Cell Dev Biol. 2025 Mar 5;13:1537505. doi: 10.3389/fcell.2025.1537505. eCollection 2025. Front Cell Dev Biol. 2025. PMID: 40109362 Free PMC article.
-
[The value of genetic diagnosis of deafness in evaluating the prognosis of cochlear implantation].Lin Chuang Er Bi Yan Hou Tou Jing Wai Ke Za Zhi. 2021 Mar;35(3):274-281. doi: 10.13201/j.issn.2096-7993.2021.03.020. Lin Chuang Er Bi Yan Hou Tou Jing Wai Ke Za Zhi. 2021. PMID: 33794619 Free PMC article. Chinese.
References
-
- Fraser GR. Association of congenital deafness with goitre (Pendred's syndrome) a study of 207 families. Ann Hum Genet. 1965;28:201–249. - PubMed
-
- Reardon W, Coffey R, Phelps PD, Luxon LM, Stephens D, et al. Pendred syndrome–100 years of underascertainment? QJM. 1997;90:443–447. - PubMed
-
- Albert S, Blons H, Jonard L, Feldmann D, Chauvin P, et al. SLC26A4 gene is frequently involved in nonsyndromic hearing impairment with enlarged vestibular aqueduct in Caucasian populations. Eur J Hum Genet. 2006;14:773–779. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

