Injury-induced GR-1+ macrophage expansion and activation occurs independently of CD4 T-cell influence
- PMID: 21430603
- PMCID: PMC3139815
- DOI: 10.1097/SHK.0b013e31821af669
Injury-induced GR-1+ macrophage expansion and activation occurs independently of CD4 T-cell influence
Abstract
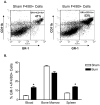
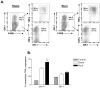
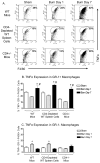
Burn injury initiates an enhanced inflammatory condition referred to as the systemic inflammatory response syndrome or the two-hit response phenotype. Prior reports indicated that macrophages respond to injury and demonstrate a heightened reactivity to Toll-like receptor stimulation. Since we and others observed a significant increase in splenic GR-1 F4/80 CD11b macrophages in burn-injured mice, we wished to test if these macrophages might be the primary macrophage subset that shows heightened LPS reactivity. We report here that burn injury promoted higher level TNF-α expression in GR-1, but not GR-1 macrophages, after LPS activation both in vivo and ex vivo. We next tested whether CD4 T cells, which are known to suppress injury-induced inflammatory responses, might control the activation and expansion of GR-1 macrophages. Interestingly, we found that GR-1 macrophage expansion and LPS-induced TNF-α expression were not significantly different between wild-type and CD4 T cell-deficient CD4(-/-) mice. However, further investigations showed that LPS-induced TNF-α production was significantly influenced by CD4 T cells. Taken together, these data indicate that GR-1 F4/80 CD11b macrophages represent the primary macrophage subset that expands in response to burn injury and that CD4 T cells do not influence the GR-1 macrophage expansion process, but do suppress LPS-induced TNF-α production. These data suggest that modulating GR-1 macrophage activation as well as CD4 T cell responses after severe injury may help control the development of systemic inflammatory response syndrome and the two-hit response phenotype.
Conflict of interest statement
Figures

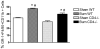

Similar articles
-
Injury primes the innate immune system for enhanced Toll-like receptor reactivity.J Immunol. 2003 Aug 1;171(3):1473-83. doi: 10.4049/jimmunol.171.3.1473. J Immunol. 2003. PMID: 12874240
-
CD4+CD25+ regulatory T cells control innate immune reactivity after injury.J Immunol. 2005 Mar 1;174(5):2957-63. doi: 10.4049/jimmunol.174.5.2957. J Immunol. 2005. PMID: 15728508
-
Linking the "two-hit" response following injury to enhanced TLR4 reactivity.J Leukoc Biol. 2005 Jan;77(1):16-23. doi: 10.1189/jlb.0704382. Epub 2004 Oct 20. J Leukoc Biol. 2005. PMID: 15496450
-
[Heme oxygenase-1 (HO-1) gene knockout affects the balance of lung immune cell composition and aggravates inflammatory injury in lung tissues of LPS-induced acute lung injury (ALI) mice].Xi Bao Yu Fen Zi Mian Yi Xue Za Zhi. 2024 Apr;40(4):296-302. Xi Bao Yu Fen Zi Mian Yi Xue Za Zhi. 2024. PMID: 38710513 Chinese.
-
Neutrophils, not monocyte/macrophages, are the major splenic source of postburn IL-10.Shock. 2011 Aug;36(2):149-55. doi: 10.1097/SHK.0b013e3182205cbc. Shock. 2011. PMID: 21558983
Cited by
-
Immune system phenotyping of radiation and radiation combined injury in outbred mice.Radiat Res. 2013 Jan;179(1):101-12. doi: 10.1667/RR3120.1. Epub 2012 Dec 5. Radiat Res. 2013. PMID: 23216446 Free PMC article.
-
Tuftsin signals through its receptor neuropilin-1 via the transforming growth factor beta pathway.J Neurochem. 2013 Nov;127(3):394-402. doi: 10.1111/jnc.12404. Epub 2013 Sep 18. J Neurochem. 2013. PMID: 24033337 Free PMC article.
-
SOCS3 Suppression Promoted the Recruitment of CD11b+Gr-1-F4/80-MHCII- Early-Stage Myeloid-Derived Suppressor Cells and Accelerated Interleukin-6-Related Tumor Invasion via Affecting Myeloid Differentiation in Breast Cancer.Front Immunol. 2018 Jul 23;9:1699. doi: 10.3389/fimmu.2018.01699. eCollection 2018. Front Immunol. 2018. PMID: 30083161 Free PMC article.
-
Gamma delta T cells regulate wound myeloid cell activity after burn.Shock. 2014 Aug;42(2):133-41. doi: 10.1097/SHK.0000000000000176. Shock. 2014. PMID: 24667623 Free PMC article.
-
Human TH9 cells are skin-tropic and have autocrine and paracrine proinflammatory capacity.Sci Transl Med. 2014 Jan 15;6(219):219ra8. doi: 10.1126/scitranslmed.3007828. Sci Transl Med. 2014. PMID: 24431112 Free PMC article.
References
-
- Lederer JA, Rodrick ML, Mannick JA. The effects of injury on the adaptive immune response. Shock. 1999;11(3):153–159. - PubMed
-
- Zedler S, Faist E. The impact of endogenous triggers on trauma-associated inflammation. Curr Opin Crit Care. 2006;12(6):595–601. - PubMed
-
- Maung AA, Fujimi S, MacConmara MP, Tajima G, McKenna AM, Delisle AJ, Stallwood C, Onderdonk AB, Mannick JA, Lederer JA. Injury enhances resistance to Escherichia coli infection by boosting innate immune system function. J Immunol. 2008;180(4):2450–2458. - PubMed
-
- O’Sullivan ST, Lederer JA, Horgan AF, Chin DH, Mannick JA, Rodrick ML. Major injury leads to predominance of the T helper-2 lymphocyte phenotype and diminished interleukin-12 production associated with decreased resistance to infection. Ann Surg. 1995;222(4):482–490. discussion 490-482. - PMC - PubMed
-
- Ni Choileain N, MacConmara M, Zang Y, Murphy TJ, Mannick JA, Lederer JA. Enhanced regulatory T cell activity is an element of the host response to injury. J Immunol. 2006;176(1):225–236. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

