Design, synthesis and biological characterization of novel inhibitors of CD38
- PMID: 21431168
- PMCID: PMC3357394
- DOI: 10.1039/c0ob00768d
Design, synthesis and biological characterization of novel inhibitors of CD38
Abstract
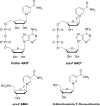
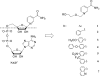
Human CD38 is a novel multi-functional protein that acts not only as an antigen for B-lymphocyte activation, but also as an enzyme catalyzing the synthesis of a Ca(2+) messenger molecule, cyclic ADP-ribose, from NAD(+). It is well established that this novel Ca(2+) signaling enzyme is responsible for regulating a wide range of physiological functions. Based on the crystal structure of the CD38/NAD(+) complex, we synthesized a series of simplified N-substituted nicotinamide derivatives (Compound 1-14). A number of these compounds exhibited moderate inhibition of the NAD(+) utilizing activity of CD38, with Compound 4 showing the highest potency. The crystal structure of CD38/Compound 4 complex and computer simulation of Compound 7 docking to CD38 show a significant role of the nicotinamide moiety and the distal aromatic group of the compounds for substrate recognition by the active site of CD38. Biologically, we showed that both Compounds 4 and 7 effectively relaxed the agonist-induced contraction of muscle preparations from rats and guinea pigs. This study is a rational design of inhibitors for CD38 that exhibit important physiological effects, and can serve as a model for future drug development.
Figures


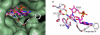
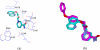




Similar articles
-
CD38 Structure-Based Inhibitor Design Using the N1-Cyclic Inosine 5'-Diphosphate Ribose Template.PLoS One. 2013 Jun 19;8(6):e66247. doi: 10.1371/journal.pone.0066247. Print 2013. PLoS One. 2013. PMID: 23840430 Free PMC article.
-
Design, synthesis and SAR studies of NAD analogues as potent inhibitors towards CD38 NADase.Molecules. 2014 Sep 29;19(10):15754-67. doi: 10.3390/molecules191015754. Molecules. 2014. PMID: 25268725 Free PMC article.
-
Rational Design and Identification of Small-Molecule Allosteric Inhibitors of CD38.Chembiochem. 2019 Oct 1;20(19):2485-2493. doi: 10.1002/cbic.201900169. Epub 2019 Aug 30. Chembiochem. 2019. PMID: 31081167
-
Ectocellular CD38-catalyzed synthesis and intracellular Ca(2+)-mobilizing activity of cyclic ADP-ribose.Cell Biochem Biophys. 1998;28(1):45-62. doi: 10.1007/BF02738309. Cell Biochem Biophys. 1998. PMID: 9386892 Review.
-
Structure and enzymology of ADP-ribosyl cyclases: conserved enzymes that produce multiple calcium mobilizing metabolites.Curr Mol Med. 2004 May;4(3):249-61. doi: 10.2174/1566524043360708. Curr Mol Med. 2004. PMID: 15101683 Review.
Cited by
-
NAD Analogs in Aid of Chemical Biology and Medicinal Chemistry.Molecules. 2019 Nov 19;24(22):4187. doi: 10.3390/molecules24224187. Molecules. 2019. PMID: 31752261 Free PMC article. Review.
-
Sirtuins as regulators of metabolism and healthspan.Nat Rev Mol Cell Biol. 2012 Mar 7;13(4):225-238. doi: 10.1038/nrm3293. Nat Rev Mol Cell Biol. 2012. PMID: 22395773 Free PMC article. Review.
-
Airway responsiveness in CD38-deficient mice in allergic airway disease: studies with bone marrow chimeras.Am J Physiol Lung Cell Mol Physiol. 2015 Mar 1;308(5):L485-93. doi: 10.1152/ajplung.00227.2014. Epub 2015 Jan 9. Am J Physiol Lung Cell Mol Physiol. 2015. PMID: 25575514 Free PMC article.
-
Stimulation of natural killer cells with small molecule inhibitors of CD38 for the treatment of neuroblastoma.Chem Sci. 2023 Jan 30;14(8):2168-2182. doi: 10.1039/d2sc05749b. eCollection 2023 Feb 22. Chem Sci. 2023. PMID: 36845935 Free PMC article.
-
CD38 in the pathogenesis of allergic airway disease: Potential therapeutic targets.Pharmacol Ther. 2017 Apr;172:116-126. doi: 10.1016/j.pharmthera.2016.12.002. Epub 2016 Dec 7. Pharmacol Ther. 2017. PMID: 27939939 Free PMC article. Review.
References
-
- Lee HC. Enzymatic functions and structures of CD38 and homologs. Chem. Immunol. 2000;75:39–59. - PubMed
-
- Lee HC. Physiological functions of cyclic ADP-ribose and NAADP as calcium messengers. Ann. Rev. Pharmacol. Toxicol. 2001;41:317–345. - PubMed
- Lee HC, Aarhus R, Levitt D. The crystal structure of cyclic ADP-ribose. Nat. Struct. Biol. 1994;1:143–144. - PubMed
- Lee HC, Walseth TF, Bratt GT, et al. Structural determination of a cyclic metabolite of NAD+ with intracellular Ca2+-mobilizing activity. J. Biol. Chem. 1989;264:1608–15. - PubMed
-
- Howard M, Grimaldi JC, Bazan JF, Lund FE, Santos-Argumedo L, Parkhouse RM, Walseth TF, Lee HC. Formation and hydrolysis of cyclic ADP-ribose catalyzed by lymphocyte antigen CD38. Science. 1993;262:1056–1059. - PubMed
-
- Kato I, Yamamoto Y, Fujimura M, Noguchi N, Takasawa S, Okamoto H. CD38 disruption impairs glucose-induced increases in Cyclic ADP-ribose, [Ca2+]i, and insulin secretion. J. Biol. Chem. 1999;274:1869–1872. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

