Targeting neurons of rat nucleus tractus solitarii with the gene transfer vector adeno-associated virus type 2 to up-regulate neuronal nitric oxide synthase
- PMID: 21431420
- PMCID: PMC3146586
- DOI: 10.1007/s10571-011-9674-5
Targeting neurons of rat nucleus tractus solitarii with the gene transfer vector adeno-associated virus type 2 to up-regulate neuronal nitric oxide synthase
Abstract
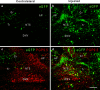
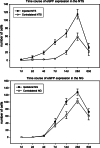
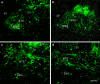
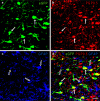
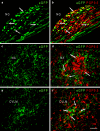
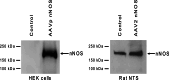
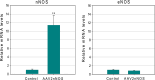
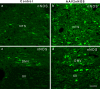
Adeno-associated virus (AAV) has distinct advantages over other viral vectors in delivering genes of interest to the brain. AAV mainly transfects neurons, produces no toxicity or inflammatory responses, and yields long-term transgene expression. In this study, we first tested the hypothesis that AAV serotype 2 (AAV2) selectively transfects neurons but not glial cells in the nucleus tractus solitarii (NTS) by examining expression of the reporter gene, enhanced green fluorescent protein (eGFP), in the rat NTS after unilateral microinjection of AAV2eGFP into NTS. Expression of eGFP was observed in 1-2 cells in the NTS 1 day after injection. The number of transduced cells and the intensity of eGFP fluorescence increased from day 1 to day 28 and decreased on day 60. The majority (92.9 ± 7.0%) of eGFP expressing NTS cells contained immunoreactivity for the neuronal marker, protein gene product 9.5, but not that for the glial marker, glial fibrillary acidic protein. We observed eGFP expressing neurons and fibers in the nodose ganglia (NG) both ipsilateral and contralateral to the injection. In addition, eGFP expressing fibers were present in both ipsilateral and contralateral nucleus ambiguus (NA), caudal ventrolateral medulla (CVLM) and rostral ventrolateral medulla (RVLM). Having established that AAV2 was able to transduce a gene into NTS neurons, we constructed AAV2 vectors that contained cDNA for neuronal nitric oxide synthase (nNOS) and examined nNOS expression in the rat NTS after injection of this vector into the area. Results from RT-PCR, Western analysis, and immunofluorescent histochemistry indicated that nNOS expression was elevated in rat NTS that had been injected with AAV2nNOS vectors. Therefore, we conclude that AAV2 is an effective viral vector in chronically transducing NTS neurons and that AAV2nNOS can be used as a specific gene transfer tool to study the role of nNOS in CNS neurons.
Figures
Similar articles
-
Feline immunodeficiency virus as a gene transfer vector in the rat nucleus tractus solitarii.Cell Mol Neurobiol. 2010 Apr;30(3):339-46. doi: 10.1007/s10571-009-9456-5. Epub 2009 Sep 24. Cell Mol Neurobiol. 2010. PMID: 19777342 Free PMC article.
-
Identification and localization of cell types that express endothelial and neuronal nitric oxide synthase in the rat nucleus tractus solitarii.Brain Res. 2007 Sep 26;1171:42-51. doi: 10.1016/j.brainres.2007.07.057. Epub 2007 Aug 8. Brain Res. 2007. PMID: 17761150 Free PMC article.
-
Nitroxidergic neurons in rat nucleus tractus solitarii express vesicular glutamate transporter 3.J Chem Neuroanat. 2005 May;29(3):179-91. doi: 10.1016/j.jchemneu.2005.01.002. J Chem Neuroanat. 2005. PMID: 15820620
-
Soluble guanylate cyclase and neuronal nitric oxide synthase colocalize in rat nucleus tractus solitarii.J Chem Neuroanat. 2005 Mar;29(2):127-36. doi: 10.1016/j.jchemneu.2004.10.002. J Chem Neuroanat. 2005. PMID: 15652699
-
Glutamatergic neurons say NO in the nucleus tractus solitarii.J Chem Neuroanat. 2009 Nov;38(3):154-65. doi: 10.1016/j.jchemneu.2009.02.002. Epub 2009 Feb 21. J Chem Neuroanat. 2009. PMID: 19778681 Free PMC article. Review.
Cited by
-
No evidence of a role for neuronal nitric oxide synthase in the nucleus tractus solitarius in ventilatory responses to acute or chronic hypoxia in awake rats.J Appl Physiol (1985). 2015 Mar 15;118(6):750-9. doi: 10.1152/japplphysiol.00333.2014. Epub 2015 Jan 8. J Appl Physiol (1985). 2015. PMID: 25571988 Free PMC article.
-
Selection of a novel cell-internalizing RNA aptamer specific for CD22 antigen in B cell acute lymphoblastic leukemia.Mol Ther Nucleic Acids. 2023 Jul 28;33:698-712. doi: 10.1016/j.omtn.2023.07.028. eCollection 2023 Sep 12. Mol Ther Nucleic Acids. 2023. PMID: 37662970 Free PMC article.
-
Cellular Localization of Acid-Sensing Ion Channel 1 in Rat Nucleus Tractus Solitarii.Cell Mol Neurobiol. 2018 Jan;38(1):219-232. doi: 10.1007/s10571-017-0534-9. Epub 2017 Aug 20. Cell Mol Neurobiol. 2018. PMID: 28825196 Free PMC article.
-
Decreased expression of neuronal nitric oxide synthase in the nucleus tractus solitarii inhibits sympathetically mediated baroreflex responses in rat.J Physiol. 2012 Aug 1;590(15):3545-59. doi: 10.1113/jphysiol.2012.237966. Epub 2012 Jun 11. J Physiol. 2012. PMID: 22687614 Free PMC article.
-
Acid-sensing ion channel 1 and nitric oxide synthase are in adjacent layers in the wall of rat and human cerebral arteries.J Chem Neuroanat. 2014 Nov;61-62:161-8. doi: 10.1016/j.jchemneu.2014.10.002. Epub 2014 Oct 23. J Chem Neuroanat. 2014. PMID: 25462386 Free PMC article.
References
-
- Allen AM, Dosanjh JK, Erac M, Dassanayake S, Hannan RD, Thomas WG (2006) Expression of constitutively active angiotensin receptors in the rostral ventrolateral medulla increases blood pressure. Hypertension 47:1054–1061 - PubMed
-
- Belur LR, Kaemmerer WF, McIvor RS, Low WC (2008) Adeno-associated virus type 2 vectors: transduction and long-term expression in cerebellar Purkinje cells in vivo is mediated by the fibroblast growth factor receptor 1: bFGFR-1 mediates AAV2 transduction of Purkinje cells. Arch Virol 153:2107–2110 - PMC - PubMed
-
- Boscan P, Pickering AE, Paton JF (2002) The nucleus of the solitary tract: an integrating station for nociceptive and cardiorespiratory afferents. Exp Physiol 87:259–266 - PubMed
-
- Boulis NM, Turner DE, Dice JA, Bhatia V, Feldman EL (1999) Characterization of adenoviral gene expression in spinal cord after remote vector delivery. Neurosurgery 45:131–137 - PubMed
-
- Bradley RM, King MS, Wang L, Shu X (1996) Neurotransmitter and neuromodulator activity in the gustatory zone of the nucleus tractus solitarius. Chem Senses 21:377–385 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

