High susceptibility of c-KIT+CD34+ precursors to prolonged doxorubicin exposure interferes with Langerhans cell differentiation in a human cell line model
- PMID: 21431918
- PMCID: PMC3119330
- DOI: 10.1007/s00262-011-1003-9
High susceptibility of c-KIT+CD34+ precursors to prolonged doxorubicin exposure interferes with Langerhans cell differentiation in a human cell line model
Abstract
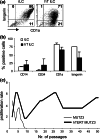
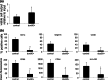
As neoadjuvant and adjuvant chemotherapy schedules often consist of multiple treatment cycles over relatively long periods of time, it is important to know what effects protracted drug administration can have on the immune system. Here, we studied the long-term effects of doxorubicin on the capacity of dendritic cell (DC) precursors to differentiate into a particular DC subset, the Langerhans cells (LC). In order to achieve high telomerase activity as detected in hematological stem cells, precursor cells from the acute-myeloid leukemia (AML)-derived cell line MUTZ3 were stably transduced with human telomerase reverse transcriptase (hTERT) to facilitate their growth potential, while preventing growth, and drug-induced senescence, and preserving their unique capacity for cytokine-dependent DC and LC differentiation. The hTERT-MUTZ3 cells were selected with increasing concentrations of the anthracyclin doxorubicin. After 1-2 months of selection with 30-90 nM doxorubicin, the cells completely lost their capacity to differentiate into LC. This inhibition turned out to be reversible, as the cells slowly regained their capacity to differentiate after a 3- to 4-month drug-free period and with this became capable again of priming allogeneic T cells. Of note, the loss and gain of this capacity to differentiate coincided with the loss and gain of a subpopulation within the CD34(+) proliferative compartment with surface expression of the stem cell factor receptor (SCF-R/CD117/c-Kit). These data are in favor of cytostatic drug-free intervals before applying autologous DC-based vaccination protocols, as specific DC precursors may need time to recover from protracted chemotherapy treatment and re-emerge among the circulating CD34(+) hematopoietic stem and precursor cells.
Conflict of interest statement
The authors have no financial conflict of interest.
Figures



Similar articles
-
Preferential Langerhans cell differentiation from CD34(+) precursors upon introduction of ABCG2 (BCRP).Immunol Cell Biol. 2012 Feb;90(2):206-15. doi: 10.1038/icb.2011.25. Epub 2011 Mar 29. Immunol Cell Biol. 2012. PMID: 21445088
-
Expression of Flt3 and c-kit during growth and maturation of human CD34+CD38- cells.Exp Hematol. 1999 May;27(5):916-27. doi: 10.1016/s0301-472x(99)00020-x. Exp Hematol. 1999. PMID: 10340408
-
Exposure of CD34+ precursors to cytostatic anthraquinone-derivatives induces rapid dendritic cell differentiation: implications for cancer immunotherapy.Cancer Immunol Immunother. 2012 Feb;61(2):181-191. doi: 10.1007/s00262-011-1039-x. Epub 2011 Aug 27. Cancer Immunol Immunother. 2012. PMID: 21874304 Free PMC article.
-
Respective involvement of TGF-beta and IL-4 in the development of Langerhans cells and non-Langerhans dendritic cells from CD34+ progenitors.J Leukoc Biol. 1999 Nov;66(5):781-91. doi: 10.1002/jlb.66.5.781. J Leukoc Biol. 1999. PMID: 10577510
-
Stem cell factor as a survival and growth factor in human normal and malignant hematopoiesis.Acta Haematol. 1996;95(3-4):257-62. doi: 10.1159/000203893. Acta Haematol. 1996. PMID: 8677752 Review.
Cited by
-
The effects of chemotherapeutic drugs on human monocyte-derived dendritic cell differentiation and antigen presentation.Clin Exp Immunol. 2013 Jun;172(3):490-9. doi: 10.1111/cei.12060. Clin Exp Immunol. 2013. PMID: 23600838 Free PMC article.
-
Comparison of the immunomodulatory potential of platinum-based anti-cancer drugs and anthracyclins on human monocyte-derived cells.Cancer Chemother Pharmacol. 2023 Jan;91(1):53-66. doi: 10.1007/s00280-022-04497-1. Epub 2022 Nov 30. Cancer Chemother Pharmacol. 2023. PMID: 36451019 Free PMC article.
References
-
- Bene MC, Bernier M, Casasnovas RO, et al. The reliability and specificity of c-kit for the diagnosis of acute myeloid leukemias and undifferentiated leukemias. The European group for the Immunological classification of leukemias (EGIL) Blood. 1998;92:596–599. - PubMed
-
- Bijl J, van Oostveen JW, Kreike M, et al. Expression of HOXC4, HOXC5, and HOXC6 in human lymphoid cell lines, leukemias, and benign and malignant lymphoid tissue. Blood. 1996;87:1737–1745. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

