Aerobic training increases skin perfusion by a nitric oxide mechanism in type 2 diabetes
- PMID: 21437095
- PMCID: PMC3047977
- DOI: 10.2147/dmsott.s12136
Aerobic training increases skin perfusion by a nitric oxide mechanism in type 2 diabetes
Abstract
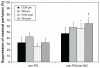
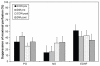
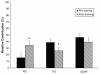
It is well known that a number of locally released vasodilatory and vasoconstrictive compounds can affect skin perfusion. This study investigated the effects of aerobic training on the contribution of nitric oxide (NO), prostaglandins (PG), and endothelial-derived hyperpolarizing factor (EDHF) in stimulated dorsal foot skin perfusion in individuals with type 2 diabetes (T2DM). Ten previously sedentary, older individuals with T2DM (57.0 ± 3.1 years) and nine sedentary controls (53.5 ± 3.2 years) were tested before and after undertaking six months of moderate aerobic training three times weekly in a supervised setting. All subjects underwent measurement of baseline (32°C) and heat-stimulated (40°C and 44°C) dorsal foot skin perfusion starting one hour after ingestion of a single, oral 325 mg dose of aspirin, a known inhibitor of PG synthesis. Before aspirin ingestion, a subcutaneous microdialysis probe was inserted into each foot dorsum to administer either saline (PG pathway only blocked by aspirin in the left foot) or L-NAME (N(G)-nitro-l-arginine methyl ester; thereby inhibiting both PG and NO pathways in the right foot). Normative data collected previously on subjects undergoing saline administration via microdialysis without aspirin ingestion served as a control group. Significantly lower responsiveness of maximal perfusion was found with the EDHF pathway alone unblocked compared with NO and EDHF unblocked after training. Maximal suppression attributable directly to NO, PG, and EDHF was not significantly different when examined by subject group and training status. However, contributions of NO, PG, and EDHF to maximal perfusion were significantly increased, decreased, and unchanged by aerobic training, respectively, with diabetic and control subjects combined due to nonsignificant differences between groups. Improvements in maximally stimulated dorsal foot skin perfusion resulting from six months of aerobic training appear to have primarily an NO basis, with lesser contributions from PG following training, regardless of diabetes status.
Keywords: aerobic training; endothelial-derived hyperpolarizing factor; microdialysis; nitric oxide; prostaglandins; skin perfusion; type 2 diabetes.
Figures
References
-
- Gooding KM, Hannemann MM, Tooke JE, Clough GF, Shore AC. Maximum skin hyperaemia induced by local heating: Possible mechanisms. J Vasc Res. 2006;43:270–277. - PubMed
-
- Lenasi H, Struci M. The effect of nitric oxide synthase and cyclooxygenase inhibition on cutaneous microvascular reactivity. Eur J Appl Physiol. 2008;103:719–726. - PubMed
-
- Sokolnicki LA, Roberts SK, Wilkins BW, Basu A, Charkoudian N. Contribution of nitric oxide to cutaneous microvascular dilation in individuals with type 2 diabetes mellitus. Am J Physiol. 2007;292:E31–E38. - PubMed
-
- Vinik AI, Erbas T, Park TS, Pierce KK, Stansberry KB. Methods for evaluation of peripheral neurovascular dysfunction. Diabetes Technol Ther. 2001;3:29–50. - PubMed
-
- Wang JS. Effects of exercise training and detraining on cutaneous microvascular function in man: The regulatory role of endothelium-dependent dilation in skin vasculature. Eur J Appl Physiol. 2005;93:429–434. - PubMed
LinkOut - more resources
Full Text Sources

