Comparison of methods for the purification of alpha-1 acid glycoprotein from human plasma
- PMID: 21437215
- PMCID: PMC3061221
- DOI: 10.1155/2011/578207
Comparison of methods for the purification of alpha-1 acid glycoprotein from human plasma
Abstract
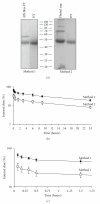
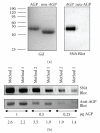
Alpha-1 acid glycoprotein (AGP) is a highly glycosylated, negatively charged plasma protein suggested to have anti-inflammatory and/or immunomodulatory activities. Purification of AGP could be simplified if methods that exploit its high solubility under chemically harsh conditions could be demonstrated to leave the protein in its native conformation. Procedures involving exposure of AGP to hot phenol or sulphosalicylic acid (SSA) were compared to solely chromatographic methods. Hot phenol-purified AGP was more rapidly cleared from mice in vivo following intravenous injection than chromatographically purified AGP. In contrast, SSA-purified AGP demonstrated an identical in vivo clearance profile and circular dichroism spectrum to chromatographically purified AGP. Similarly, no differences in susceptibility to enzymatic deglycosylation or reactivity with Sambucus nigra lectin were detected between AGP purified via the two methods. Incorporation of the SSA step in the purification scheme for AGP eliminated the need for a large (4 mL resin/mL of plasma) initial chromatographic step and simplified its purification without causing any detectable distortion in the conformation of the protein. Confirmation that this procedure is nondenaturing will simplify AGP purification and investigation of its possible biological roles in laboratory animals.
Figures


Similar articles
-
One-step purification of rat plasma alpha 1-acid glycoprotein by antibody affinity chromatography: application to normal and inflamed rat sera.J Chromatogr. 1989 Apr 14;489(2):273-81. doi: 10.1016/s0378-4347(00)82905-9. J Chromatogr. 1989. PMID: 2753951
-
In vivo clearance of alpha-1 acid glycoprotein is influenced by the extent of its N-linked glycosylation and by its interaction with the vessel wall.J Biomed Biotechnol. 2012;2012:292730. doi: 10.1155/2012/292730. Epub 2012 Apr 1. J Biomed Biotechnol. 2012. PMID: 22545002 Free PMC article.
-
Heterogeneity of alpha 1-acid glycoprotein in rheumatoid arthritis.J Chromatogr B Biomed Appl. 1994 Nov 4;661(1):7-14. doi: 10.1016/0378-4347(94)00335-1. J Chromatogr B Biomed Appl. 1994. PMID: 7866554
-
Alpha-1-acid glycoprotein.Biochim Biophys Acta. 2000 Oct 18;1482(1-2):157-71. doi: 10.1016/s0167-4838(00)00153-9. Biochim Biophys Acta. 2000. PMID: 11058758 Review.
-
The Immune Functions of α1 Acid Glycoprotein.Curr Protein Pept Sci. 2019;20(6):505-524. doi: 10.2174/1389203720666190405101138. Curr Protein Pept Sci. 2019. PMID: 30950347 Review.
Cited by
-
Serum Levels and Glycosylation Changes of Alpha-1-Acid Glycoprotein According to Severity of Breast Cancer in Korean Women.J Microbiol Biotechnol. 2020 Sep 28;30(9):1297-1304. doi: 10.4014/jmb.2006.06007. J Microbiol Biotechnol. 2020. PMID: 32627751 Free PMC article.
-
Development of a novel system for mass spectrometric analysis of cancer-associated fucosylation in plasma α1-acid glycoprotein.Biomed Res Int. 2013;2013:834790. doi: 10.1155/2013/834790. Epub 2013 Feb 13. Biomed Res Int. 2013. PMID: 23509786 Free PMC article.
-
Serum biomarker discovery related to pathogenesis in acute coronary syndrome by proteomic approach.Biosci Rep. 2021 Jun 25;41(6):BSR20210344. doi: 10.1042/BSR20210344. Biosci Rep. 2021. PMID: 34002800 Free PMC article.
-
Hyaluronic Acid Coated Chitosan Nanoparticles Reduced the Immunogenicity of the Formed Protein Corona.Sci Rep. 2017 Sep 5;7(1):10542. doi: 10.1038/s41598-017-10836-7. Sci Rep. 2017. PMID: 28874846 Free PMC article.
References
-
- Schmid K, Nimberg RB, Kimura A, et al. The carbohydrate units of human plasma α1-acid glycoprotein. Biochimica et Biophysica Acta. 1977;492(2):291–302. - PubMed
-
- Fournier T, Medjoubi N, Porquet D. Alpha-1-acid glycoprotein. Biochimica et Biophysica Acta. 2000;1482(1-2):157–171. - PubMed
-
- Nicollet I, Lebreton JP, Fontaine M, et al. Evidence for alpha-1-acid glycoprotein populations of different pI values after concanavalin A affinity chromatography. Study of their evolution during inflammation in man. Biochimica et Biophysica Acta. 1981;668:235–245. - PubMed
-
- Schönfeld DL, Ravelli RBG, Mueller U, Skerra A. The 1.8-Å crystal structure of α1-acid glycoprotein (orosomucoid) solved by UV RIP reveals the broad drug-binding activity of this human plasma lipocalin. Journal of Molecular Biology. 2008;384(2):393–405. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

