Tissue destruction and invasion by Entamoeba histolytica
- PMID: 21440507
- PMCID: PMC3104091
- DOI: 10.1016/j.pt.2011.02.006
Tissue destruction and invasion by Entamoeba histolytica
Abstract
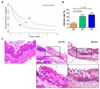
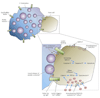
Entamoeba histolytica is the causative agent of amebiasis, a disease that is a major source of morbidity and mortality in the developing world. The potent cytotoxic activity of the parasite appears to underlie disease pathogenesis, although the mechanism is unknown. Recently, progress has been made in determining that the parasite activates apoptosis in target cells and some putative effectors have been identified. Recent studies have also begun to unravel the host genetic determinants that influence infection outcome. Thus, we are beginning to get a clearer picture of how this parasite manages to infect, invade and ultimately inflict devastating tissue destruction.
Figures


References
-
- WHO/PAHO/UNESCO report. A consultation with experts on amoebiasis. Mexico City, Mexico 28–29 January, 1997. Epidemiol Bull. 1997;18:13–14. - PubMed
-
- Mondal D, et al. Entamoeba histolytica-associated diarrheal illness is negatively associated with the growth of preschool children: evidence from a prospective study. Trans R Soc Trop Med Hyg. 2006;100:1032–1038. - PubMed
-
- Petri WA, Jr, et al. The bittersweet interface of parasite and host: lectin-carbohydrate interactions during human invasion by the parasite Entamoeba histolytica. Annu Rev Microbiol. 2002;56:39–64. - PubMed
-
- Petri WA., Jr Therapy of intestinal protozoa. Trends Parasitol. 2003;19:523–526. - PubMed
-
- Pritt BS, Clark CG. Amebiasis. Mayo Clin Proc. 2008;83:1154–1159. quiz 1159–1160. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

