A lipocentric view of peptide-induced pores
- PMID: 21442255
- PMCID: PMC3070086
- DOI: 10.1007/s00249-011-0693-4
A lipocentric view of peptide-induced pores
Abstract
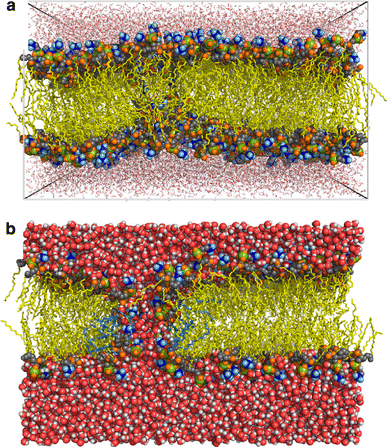
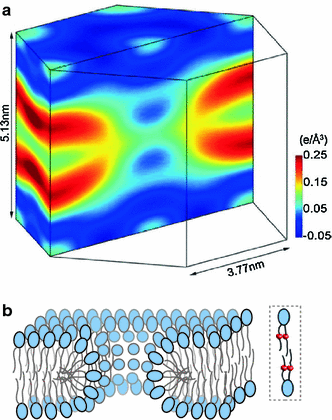
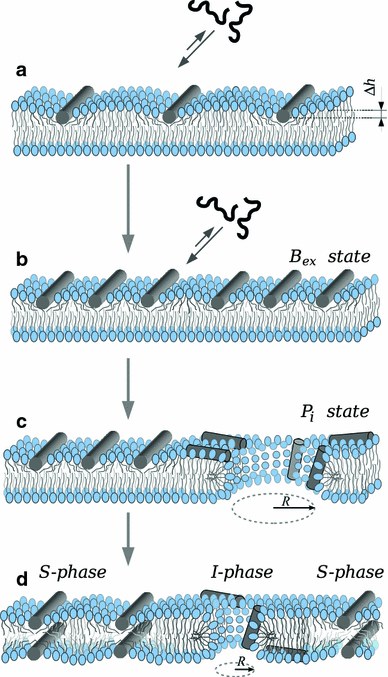
Although lipid membranes serve as effective sealing barriers for the passage of most polar solutes, nonmediated leakage is not completely improbable. A high activation energy normally keeps unassisted bilayer permeation at a very low frequency, but lipids are able to self-organize as pores even in peptide-free and protein-free membranes. The probability of leakage phenomena increases under conditions such as phase coexistence, external stress or perturbation associated to binding of nonlipidic molecules. Here, we argue that pore formation can be viewed as an intrinsic property of lipid bilayers, with strong similarities in the structure and mechanism between pores formed with participation of peptides, lipidic pores induced by different types of stress, and spontaneous transient bilayer defects driven by thermal fluctuations. Within such a lipocentric framework, amphipathic peptides are best described as pore-inducing rather than pore-forming elements. Active peptides bound to membranes can be understood as a source of internal surface tension which facilitates pore formation by diminishing the high activation energy barrier. This first or immediate action of the peptide has some resemblance to catalysis. However, the presence of membrane-active peptides has the additional effect of displacing the equilibrium towards the pore-open state, which is then maintained over long times, and reducing the size of initial individual pores. Thus, pore-inducing peptides, regardless of their sequence and oligomeric organization, can be assigned a double role of increasing the probability of pore formation in membranes to high levels as well as stabilizing these pores after they appear.
Figures




Similar articles
-
Role of membrane lipids for the activity of pore forming peptides and proteins.Adv Exp Med Biol. 2010;677:31-55. doi: 10.1007/978-1-4419-6327-7_4. Adv Exp Med Biol. 2010. PMID: 20687479 Review.
-
Interaction of hagfish cathelicidin antimicrobial peptides with model lipid membranes.FEBS Lett. 2002 Dec 4;532(1-2):115-20. doi: 10.1016/s0014-5793(02)03651-7. FEBS Lett. 2002. PMID: 12459474
-
Antimicrobial Peptide Simulations and the Influence of Force Field on the Free Energy for Pore Formation in Lipid Bilayers.J Chem Theory Comput. 2016 Sep 13;12(9):4524-33. doi: 10.1021/acs.jctc.6b00265. Epub 2016 Aug 30. J Chem Theory Comput. 2016. PMID: 27529120
-
Energetics and self-assembly of amphipathic peptide pores in lipid membranes.Biophys J. 2003 Apr;84(4):2242-55. doi: 10.1016/S0006-3495(03)75030-9. Biophys J. 2003. PMID: 12668433 Free PMC article.
-
Estimation of pore dimensions in lipid membranes induced by peptides and other biomolecules: A review.Biochim Biophys Acta Biomembr. 2021 Apr 1;1863(4):183551. doi: 10.1016/j.bbamem.2021.183551. Epub 2021 Jan 16. Biochim Biophys Acta Biomembr. 2021. PMID: 33465367 Review.
Cited by
-
Designing antimicrobial peptides: form follows function.Nat Rev Drug Discov. 2011 Dec 16;11(1):37-51. doi: 10.1038/nrd3591. Nat Rev Drug Discov. 2011. PMID: 22173434 Review.
-
Pore formation in regulated cell death.EMBO J. 2020 Dec 1;39(23):e105753. doi: 10.15252/embj.2020105753. Epub 2020 Oct 30. EMBO J. 2020. PMID: 33124082 Free PMC article. Review.
-
Effects of cholesterol on the anionic magnetite nanoparticle-induced deformation and poration of giant lipid vesicles.RSC Adv. 2022 Oct 4;12(44):28283-28294. doi: 10.1039/d2ra03199j. eCollection 2022 Oct 4. RSC Adv. 2022. PMID: 36320506 Free PMC article.
-
Anti-Virulence Potential of a Chionodracine-Derived Peptide against Multidrug-Resistant Pseudomonas aeruginosa Clinical Isolates from Cystic Fibrosis Patients.Int J Mol Sci. 2022 Nov 4;23(21):13494. doi: 10.3390/ijms232113494. Int J Mol Sci. 2022. PMID: 36362282 Free PMC article.
-
Recent developments in the kinetics of ruptures of giant vesicles under constant tension.RSC Adv. 2021 Sep 2;11(47):29598-29619. doi: 10.1039/d1ra04647k. eCollection 2021 Sep 1. RSC Adv. 2021. PMID: 35479542 Free PMC article. Review.
References
-
- Abidor I, Arakelyan V, Chernomordik Y, Chizmadzhev V, Tarasevich M. Electrical breakdown ofBLM: main experimental facts and their qualitative discussion. Bioelectrochem Bioenerg. 1979;6:37–52.
-
- Andersen S, Jackson A, Heimburg T. Towards a thermodynamic theory of nerve pulse propagation. Prog Neurobiol. 2009;88:104–113. - PubMed
-
- Antonov VF, Petrov VV, Molnar AA, Predvoditelev DA, Ivanov AS. The appearance of single-ion channels in unmodified lipid bilayer membranes at the phase transition temperature. Nature. 1980;283:585–586. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

