Linking lipids to Alzheimer's disease: cholesterol and beyond
- PMID: 21448224
- PMCID: PMC3321383
- DOI: 10.1038/nrn3012
Linking lipids to Alzheimer's disease: cholesterol and beyond
Erratum in
- Nat Rev Neurosci. 2011 Aug;12(8):484
Abstract
Lipid-mediated signalling regulates a plethora of physiological processes, including crucial aspects of brain function. In addition, dysregulation of lipid pathways has been implicated in a growing number of neurodegenerative disorders, such as Alzheimer's disease (AD). Although much attention has been given to the link between cholesterol and AD pathogenesis, growing evidence suggests that other lipids, such as phosphoinositides and phosphatidic acid, have an important role. Regulators of lipid metabolism (for example, statins) are a highly successful class of marketed drugs, and exploration of lipid dysregulation in AD and identification of novel therapeutic agents acting through relevant lipid pathways offers new and effective options for the treatment of this devastating disorder.
Figures
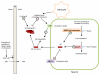
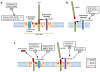
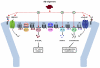
References
-
- Haass C, Selkoe DJ. Soluble protein oligomers in neurodegeneration: lessons from the Alzheimer’s amyloid beta-peptide. Nat Rev Mol Cell Biol. 2007;8:101–112. - PubMed
-
- Tanzi RE, Bertram L. Twenty years of the Alzheimer’s disease amyloid hypothesis: a genetic perspective. Cell. 2005;120:545–555. - PubMed
-
- Ballatore C, Lee VM, Trojanowski JQ. Tau-mediated neurodegeneration in Alzheimer’s disease and related disorders. Nat Rev Neurosci. 2007;8:663–672. - PubMed
-
- Foley P. Lipids in Alzheimer’s disease: A century-old story. Biochimica et biophysica acta. 2010;1801:750–753. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

