Growth hormone plus childhood low-dose estrogen in Turner's syndrome
- PMID: 21449786
- PMCID: PMC3083123
- DOI: 10.1056/NEJMoa1005669
Growth hormone plus childhood low-dose estrogen in Turner's syndrome
Abstract
Background: Short stature and ovarian failure are characteristic features of Turner's syndrome. Although recombinant human growth hormone is commonly used to treat the short stature associated with this syndrome, a randomized, placebo-controlled trial is needed to document whether such treatment increases adult height. Furthermore, it is not known whether childhood estrogen replacement combined with growth hormone therapy provides additional benefit. We examined the independent and combined effects of growth hormone and early, ultra-low-dose estrogen on adult height in girls with Turner's syndrome.
Methods: In this double-blind, placebo-controlled trial, we randomly assigned 149 girls, 5.0 to 12.5 years of age, to four groups: double placebo (placebo injection plus childhood oral placebo, 39 patients), estrogen alone (placebo injection plus childhood oral low-dose estrogen, 40), growth hormone alone (growth hormone injection plus childhood oral placebo, 35), and growth hormone-estrogen (growth hormone injection plus childhood oral low-dose estrogen, 35). The dose of growth hormone was 0.1 mg per kilogram of body weight three times per week. The doses of ethinyl estradiol (or placebo) were adjusted for chronologic age and pubertal status. At the first visit after the age of 12.0 years, patients in all treatment groups received escalating doses of ethinyl estradiol. Growth hormone injections were terminated when adult height was reached.
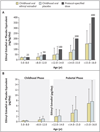
Results: The mean standard-deviation scores for adult height, attained at an average age of 17.0±1.0 years, after an average study period of 7.2±2.5 years were -2.81±0.85, -3.39±0.74, -2.29±1.10, and -2.10±1.02 for the double-placebo, estrogen-alone, growth hormone-alone, and growth hormone-estrogen groups, respectively (P<0.001). The overall effect of growth hormone treatment (vs. placebo) on adult height was a 0.78±0.13 increase in the height standard-deviation score (5.0 cm) (P<0.001); adult height was greater in the growth hormone-estrogen group than in the growth hormone-alone group, by 0.32±0.17 standard-deviation score (2.1 cm) (P=0.059), suggesting a modest synergy between childhood low-dose ethinyl estradiol and growth hormone.
Conclusions: Our study shows that growth hormone treatment increases adult height in patients with Turner's syndrome. In addition, the data suggest that combining childhood ultra-low-dose estrogen with growth hormone may improve growth and provide other potential benefits associated with early initiation of estrogen replacement. (Funded by the National Institute of Child Health and Human Development and Eli Lilly; ClinicalTrials.gov number, NCT00001221.).
Figures

Comment in
-
Assessing the value of treatments to increase height.N Engl J Med. 2011 Mar 31;364(13):1274-6. doi: 10.1056/NEJMe1012987. N Engl J Med. 2011. PMID: 21449792 No abstract available.
References
-
- Nielsen J, Wohlert M. Chromosome abnormalities found among 34,910 newborn children: results from a 13-year incidence study in Arhus, Denmark. Hum Genet. 1991;87:81–83. - PubMed
-
- Bondy CA. Care of girls and women with Turner syndrome: a guideline of the Turner Syndrome Study Group. J Clin Endocrinol Metab. 2007;92:10–25. - PubMed
-
- Davenport ML, Punyasavatsut N, Stewart PW, Gunther DF, Savendahl L, Sybert VP. Growth failure in early life: an important manifestation of Turner syndrome. Horm Res. 2002;57:157–164. - PubMed
-
- Rosenfeld RG, Attie KM, Frane J, et al. Growth hormone therapy of Turner’s syndrome: beneficial effect on adult height. J Pediatr. 1998;132:319–324. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
