Hyperphosphorylated Tau in an α-synuclein-overexpressing transgenic model of Parkinson's disease
- PMID: 21453448
- PMCID: PMC3086951
- DOI: 10.1111/j.1460-9568.2011.07660.x
Hyperphosphorylated Tau in an α-synuclein-overexpressing transgenic model of Parkinson's disease
Abstract
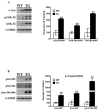
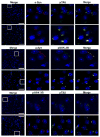
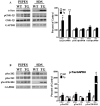
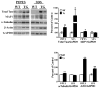
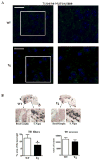
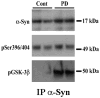
Although clinically distinct diseases, tauopathies and synucleinopathies share a common genesis and mechanisms, leading to overlapping degenerative changes within neurons. In human postmortem striatum of Parkinson's disease (PD) and PD with dementia, we have recently described elevated levels of tauopathy, indexed as increased hyperphosphorylated Tau (p-Tau). Here we assessed tauopathy in striatum of a transgenic animal model of PD, overexpressing human α-synuclein under the platelet-derived growth factor promoter. At 11 months of age, large and progressive increases in p-Tau in transgenic mice, hyperphosphorylated at sites reminiscent of Alzheimer's disease, were noted, along with elevated levels of α-synuclein and glycogen synthase kinase 3β phosphorylated at Tyr216 (p-GSK-3β), a major kinase involved in the hyperphosphorylation of Tau. Differential Triton X-100 extraction of striata showed the presence of aggregated α-synuclein in the transgenic mice, along with p-Tau and p-GSK-3β, which was also confirmed through immunohistochemistry. After p-Tau formation, both Tau and microtubule-associated protein 1 (MAP1) dissociated from the cytoskeleton, consistent with the diminished ability of these cytoskeleton-binding proteins to bind microtubules. Increases in free tubulin and actin were also noted, indicative of cytoskeleton remodeling and destabilization. In vivo magnetic resonance imaging of the transgenic animals showed a reduction in brain volume of transgenic mice, indicating substantial atrophy. From immunohistochemical studies, α-synuclein, p-Tau and p-GSK-3β were found to be overexpressed and co-localized in large inclusion bodies, reminiscent of Lewy bodies. The elevated state of tauopathy seen in these platelet-derived growth factor-α-synuclein mice provides further confirmation that PD may be a tauopathic disease.
© 2011 The Authors. European Journal of Neuroscience © 2011 Federation of European Neuroscience Societies and Blackwell Publishing Ltd.
Figures




Similar articles
-
Tauopathic changes in the striatum of A53T α-synuclein mutant mouse model of Parkinson's disease.PLoS One. 2011 Mar 21;6(3):e17953. doi: 10.1371/journal.pone.0017953. PLoS One. 2011. PMID: 21445308 Free PMC article.
-
Elevated tauopathy and alpha-synuclein pathology in postmortem Parkinson's disease brains with and without dementia.Exp Neurol. 2010 Sep;225(1):210-8. doi: 10.1016/j.expneurol.2010.06.017. Epub 2010 Jun 28. Exp Neurol. 2010. PMID: 20599975 Free PMC article.
-
Alpha-Synuclein contributes to GSK-3beta-catalyzed Tau phosphorylation in Parkinson's disease models.FASEB J. 2009 Sep;23(9):2820-30. doi: 10.1096/fj.08-120410. Epub 2009 Apr 15. FASEB J. 2009. PMID: 19369384 Free PMC article.
-
Association of glycogen synthase kinase-3β with Parkinson's disease (review).Mol Med Rep. 2014 Jun;9(6):2043-50. doi: 10.3892/mmr.2014.2080. Epub 2014 Mar 28. Mol Med Rep. 2014. PMID: 24681994 Free PMC article. Review.
-
Synaptic sabotage: How Tau and α-Synuclein undermine synaptic health.J Cell Biol. 2025 Feb 3;224(2):e202409104. doi: 10.1083/jcb.202409104. Epub 2024 Dec 24. J Cell Biol. 2025. PMID: 39718548 Review.
Cited by
-
Pathogenesis of synaptic degeneration in Alzheimer's disease and Lewy body disease.Biochem Pharmacol. 2014 Apr 15;88(4):508-16. doi: 10.1016/j.bcp.2014.01.015. Epub 2014 Jan 21. Biochem Pharmacol. 2014. PMID: 24462903 Free PMC article. Review.
-
Novel chemical inhibitor against SOD1 misfolding and aggregation protects neuron-loss and ameliorates disease symptoms in ALS mouse model.Commun Biol. 2021 Dec 15;4(1):1397. doi: 10.1038/s42003-021-02862-z. Commun Biol. 2021. PMID: 34912047 Free PMC article.
-
Dementia with Lewy bodies and Parkinson's disease-dementia: current concepts and controversies.J Neural Transm (Vienna). 2018 Apr;125(4):615-650. doi: 10.1007/s00702-017-1821-9. Epub 2017 Dec 8. J Neural Transm (Vienna). 2018. PMID: 29222591 Review.
-
Examining the Toxicity of α-Synuclein in Neurodegenerative Disorders.Life (Basel). 2021 Oct 22;11(11):1126. doi: 10.3390/life11111126. Life (Basel). 2021. PMID: 34833002 Free PMC article. Review.
-
Neuroimmune Tau Mechanisms: Their Role in the Progression of Neuronal Degeneration.Int J Mol Sci. 2018 Mar 23;19(4):956. doi: 10.3390/ijms19040956. Int J Mol Sci. 2018. PMID: 29570615 Free PMC article. Review.
References
-
- Abraha A, Ghoshal N, Gamblin TC, Cryns V, Berry RW, Kuret J, Binder LIJ. C-terminal inhibition of Tau assembly in vitro and in Alzheimer's disease. Cell Sci. 2000;113(21):3737–3745. - PubMed
-
- Alonso AD, Zaidi T, Novak M, Barra HS, Grundke-Iqbal I, Iqbal K. Interaction of Tau isoforms with Alzheimer's disease abnormally hyperphosphorylated Tau and in vitro phosphorylation into the disease-like protein. J Biol Chem. 2001;276(41):37967–73. - PubMed
-
- Corti O, Hampe C, Darios F, Ibanez P, Ruberg M, Brice A. Parkinson's disease: from causes to mechanisms. C R Biol. 2005;328(2):131–42. Review. - PubMed
-
- Dickson DW, Bergeron C, Chin SS, Duyckaerts C, Horoupian D, Ikeda K, Jellinger K, Lantos PL, Lippa CF, Mirra SS, Tabaton M, Vonsattel JP, Wakabayashi K, Litvan I. Office of Rare Diseases neuropathologic criteria for corticobasal degeneration. J Neuropathol Exp Neurol. 2002;61(11):935–46. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

