Effect of kallikrein 4 loss on enamel mineralization: comparison with mice lacking matrix metalloproteinase 20
- PMID: 21454549
- PMCID: PMC3093887
- DOI: 10.1074/jbc.M110.194258
Effect of kallikrein 4 loss on enamel mineralization: comparison with mice lacking matrix metalloproteinase 20
Abstract
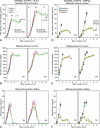
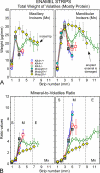
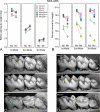
Enamel formation depends on a triad of tissue-specific matrix proteins (amelogenin, ameloblastin, and enamelin) to help initiate and stabilize progressively elongating, thin mineral ribbons of hydroxyapatite formed during an appositional growth phase. Subsequently, these proteins are eradicated to facilitate lateral expansion of the hydroxyapatite crystallites. The purpose of this study was to investigate changes in enamel mineralization occurring in mice unable to produce kallikrein 4 (Klk4), a proteinase associated with terminal extracellular degradation of matrix proteins during the maturation stage. Mice lacking functional matrix metalloproteinase 20 (Mmp20), a proteinase associated with early cleavage of matrix proteins during the secretory stage, were also analyzed as a frame of reference. The results indicated that mice lacking Klk4 produce enamel that is normal in thickness and overall organization in terms of layers and rod/inter-rod structure, but there is a developmental defect in enamel rods where they first form near the dentinoenamel junction. Mineralization is normal up to early maturation after which the enamel both retains and gains additional proteins and is unable to mature beyond 85% mineral by weight. The outmost enamel is hard, but inner regions are soft and contain much more protein than normal. The rate of mineral acquisition overall is lower by 25%. Mice lacking functional Mmp20 produce enamel that is thin and structurally abnormal. Relatively high amounts of protein remain throughout maturation, but the enamel is able to change from 67 to 75% mineral by weight during maturation. These findings reaffirm the importance of secreted proteinases to enamel mineral acquisition.
© 2011 by The American Society for Biochemistry and Molecular Biology, Inc.
Figures



Similar articles
-
Enamel proteins and proteases in Mmp20 and Klk4 null and double-null mice.Eur J Oral Sci. 2011 Dec;119 Suppl 1(Suppl 1):206-16. doi: 10.1111/j.1600-0722.2011.00866.x. Eur J Oral Sci. 2011. PMID: 22243248 Free PMC article.
-
Functions of KLK4 and MMP-20 in dental enamel formation.Biol Chem. 2008 Jun;389(6):695-700. doi: 10.1515/BC.2008.080. Biol Chem. 2008. PMID: 18627287 Free PMC article. Review.
-
Relationships between protein and mineral during enamel development in normal and genetically altered mice.Eur J Oral Sci. 2011 Dec;119 Suppl 1(Suppl 1):125-35. doi: 10.1111/j.1600-0722.2011.00871.x. Eur J Oral Sci. 2011. PMID: 22243238 Free PMC article.
-
Kallikrein-related peptidase 4, matrix metalloproteinase 20, and the maturation of murine and porcine enamel.Eur J Oral Sci. 2011 Dec;119 Suppl 1(Suppl 1):217-25. doi: 10.1111/j.1600-0722.2011.00859.x. Eur J Oral Sci. 2011. PMID: 22243249 Free PMC article.
-
Expression, structure, and function of enamel proteinases.Connect Tissue Res. 2002;43(2-3):441-9. doi: 10.1080/03008200290001159. Connect Tissue Res. 2002. PMID: 12489196 Review.
Cited by
-
Dental malformations associated with biallelic MMP20 mutations.Mol Genet Genomic Med. 2020 Aug;8(8):e1307. doi: 10.1002/mgg3.1307. Epub 2020 Jun 3. Mol Genet Genomic Med. 2020. PMID: 32495503 Free PMC article.
-
Protein-mediated enamel mineralization.Front Biosci (Landmark Ed). 2012 Jun 1;17(6):1996-2023. doi: 10.2741/4034. Front Biosci (Landmark Ed). 2012. PMID: 22652761 Free PMC article. Review.
-
MMP20 and KLK4 activation and inactivation interactions in vitro.Arch Oral Biol. 2013 Nov;58(11):1569-77. doi: 10.1016/j.archoralbio.2013.08.005. Epub 2013 Aug 18. Arch Oral Biol. 2013. PMID: 24112721 Free PMC article.
-
Evolution of Klk4 and enamel maturation in eutherians.Biol Chem. 2014 Sep;395(9):1003-13. doi: 10.1515/hsz-2014-0122. Biol Chem. 2014. PMID: 25153384 Free PMC article.
-
Genetic variation in MMP20 contributes to higher caries experience.J Dent. 2012 May;40(5):381-6. doi: 10.1016/j.jdent.2012.01.015. Epub 2012 Feb 3. J Dent. 2012. PMID: 22330321 Free PMC article.
References
-
- Hu J. C., Chun Y. H., Al Hazzazzi T., Simmer J. P. (2007) Cells Tissues Organs 186, 78–85 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

