Inherited dysfunction of sarcoplasmic reticulum Ca2+ handling and arrhythmogenesis
- PMID: 21454795
- PMCID: PMC3085083
- DOI: 10.1161/CIRCRESAHA.110.226845
Inherited dysfunction of sarcoplasmic reticulum Ca2+ handling and arrhythmogenesis
Abstract
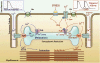
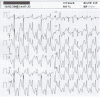
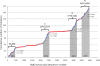
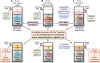
Catecholaminergic polymorphic ventricular tachycardia (CPVT) is an inherited arrhythmogenic disease occurring in patients with a structurally normal heart: the disease is characterized by life-threatening arrhythmias elicited by stress and emotion. In 2001, the ryanodine receptor was identified as the gene that is linked to CPVT; shortly thereafter, cardiac calsequestrin was implicated in the recessive form of the same disease. It became clear that abnormalities in intracellular Ca(2+) regulation could profoundly disrupt the electrophysiological properties of the heart. In this article, we discuss the molecular basis of the disease and the pathophysiological mechanisms that are impacting clinical diagnosis and management of affected individuals. As of today, the interaction between basic scientists and clinicians to understand CPVT and identify new therapeutic strategies is one of the most compelling examples of the importance of translational research in cardiology.
Figures


References
-
- Coumel P, Fidelle J, Lucet V, Attuel P, Bouvrain Y. Catecholaminergic-induced severe ventricular arrhythmias with Adams-Stokes syndrome in children: report of four cases. Br Heart J. 1978;40:28–37.
-
- Leenhardt A, Lucet V, Denjoy I, Grau F, Ngoc DD, Coumel P. Catecholaminergic polymorphic ventricular tachycardia in children. A 7-year follow-up of 21 patients. Circulation. 1995;91:1512–1519. - PubMed
-
- Priori SG, Napolitano C, Tiso N, Memmi M, Vignati G, Bloise R, Sorrentino V, Danieli GA. Mutations in the Cardiac Ryanodine Receptor Gene (hRyR2) Underlie Catecholaminergic Polymorphic Ventricular Tachycardia. Circulation. 2001;103:196–200. - PubMed
-
- Swan H, Piippo K, Viitasalo M, Heikkila P, Paavonen T, Kainulainen K, Kere J, Keto P, Kontula K, Toivonen L. Arrhythmic disorder mapped to chromosome 1q42-q43 causes malignant polymorphic ventricular tachycardia in structurally normal hearts. J Am.Coll.Cardiol. 1999;34:2035–2042. - PubMed
-
- Lahat H, Pras E, Olender T, Avidan N, Ben Asher E, Man O, Levy-Nissenbaum E, Khoury A, Lorber A, Goldman B, Lancet D, Eldar M. A missense mutation in a highly conserved region of CASQ2 is associated with autosomal recessive catecholamine-induced polymorphic ventricular tachycardia in Bedouin families from Israel. Am J Hum.Genet. 2001;69:1378–1384. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

