Abundant non-canonical dUTP found in primary human macrophages drives its frequent incorporation by HIV-1 reverse transcriptase
- PMID: 21454906
- PMCID: PMC3137078
- DOI: 10.1074/jbc.M111.234047
Abundant non-canonical dUTP found in primary human macrophages drives its frequent incorporation by HIV-1 reverse transcriptase
Abstract
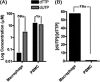
Terminally differentiated/non-dividing macrophages contain extremely low cellular dNTP concentrations (20-40 nm), compared with activated CD4(+) T cells (2-5 μm). However, our LC-MS/MS study revealed that the non-canonical dUTP concentration (2.9 μm) is ∼60 times higher than TTP in macrophages, whereas the concentrations of dUTP and TTP in dividing human primary lymphocytes are very similar. Specifically, we evaluated the contribution of HIV-1 reverse transcriptase to proviral DNA uracilation under the physiological conditions found in HIV-1 target cells. Indeed, biochemical simulation of HIV-1 reverse transcription demonstrates that HIV-1 RT efficiently incorporates dUTP in the macrophage nucleotide pools but not in the T cell nucleotide pools. Measurement of both pre-steady state and steady state kinetic parameters of dUTP incorporation reveals minimal selectivity of HIV-1 RT for TTP over dUTP, implying that the cellular dUTP/TTP ratio determines the frequency of HIV-1 RT-mediated dUTP incorporation. The RT of another lentivirus, simian immunodeficiency virus, also displays efficient dUTP incorporation in the dNTP/dUTP pools found in macrophages but not in T cells. Finally, 2',3'-dideoxyuridine was inhibitory to HIV-1 proviral DNA synthesis in macrophages but not in T cells. The data presented demonstrates that the non-canonical dUTP was abundant relative to TTP, and efficiently incorporated during HIV-1 reverse transcription, particularly in non-dividing macrophages.
Figures




References
-
- Brynolf K., Eliasson R., Reichard P. (1978) Cell 13, 573–580 - PubMed
-
- Nilsen H., Rosewell I., Robins P., Skjelbred C. F., Andersen S., Slupphaug G., Daly G., Krokan H. E., Lindahl T., Barnes D. E. (2000) Mol. Cell 5, 1059–1065 - PubMed
-
- Chen R., Wang H., Mansky L. M. (2002) J. Gen. Virol. 83, 2339–2345 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

