DIF-1 regulates Dictyostelium basal disc differentiation by inducing the nuclear accumulation of a bZIP transcription factor
- PMID: 21458438
- PMCID: PMC3107940
- DOI: 10.1016/j.ydbio.2011.03.024
DIF-1 regulates Dictyostelium basal disc differentiation by inducing the nuclear accumulation of a bZIP transcription factor
Abstract
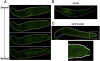
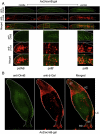
Exposure of monolayer Dictyostelium cells to the signalling polyketide DIF-1 causes DimB, a bZIPtranscription factor, to accumulate in the nucleus where it induces prestalk gene expression. Here we analyse DimB signalling during normal development. In slugs DimB is specifically nuclear enriched in the pstB cells; a cluster of vital dye-staining cells located on the ventral surface of the posterior, prespore region. PstB cells move at culmination, to form the lower cup and the outer basal disc of the fruiting body, and DimB retains a high nuclear concentration in both these tissues. In a dimB null (dimB-) strain there are very few pstB or lower cup cells, as detected by neutral red staining, and it is known that the outer basal disc is absent or much reduced. In the dimB- strain ecmB, a marker of pstB differentiation, is not DIF inducible. Furthermore, ChIP analysis shows that DimB binds to the ecmB promoter in DIF-induced cells. These results suggest that the differentiation of pstB cells is caused by a high perceived level of DIF-1 signalling, leading to nuclear localization of DimB and direct activation of cell type-specific gene expression.
Copyright © 2011 Elsevier Inc. All rights reserved.
Figures









References
-
- Brookman J.J., Jermyn K.A., Kay R.R. Nature and distribution of the morphogen DIF in the Dictyostelium slug. Development. 1987;100:119–124. - PubMed
-
- Ceccarelli A., Mahbubani H., Williams J.G. Positively and negatively acting signals regulating stalk cell and anterior-like cell differentiation in Dictyostelium. Cell. 1991;65:983–989. - PubMed
-
- Ceccarelli A., Zhukovskaya N., Kawata T., Bozzaro S., Williams J.G. Characterisation of a DNA sequence element that directs Dictyostelium stalk cell-specific gene expression. Differentiation. 2000;66:189–196. - PubMed
-
- Dingermann T. Optimization and in situ detection of Escherichia coli beta-galactosidase gene expression in Dictyostelium discoideum. Gene. 1989;85:353–362. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

