ATP hydrolysis-dependent asymmetry of the conformation of CFTR channel pore
- PMID: 21461971
- PMCID: PMC10717511
- DOI: 10.1007/s12576-011-0144-0
ATP hydrolysis-dependent asymmetry of the conformation of CFTR channel pore
Abstract
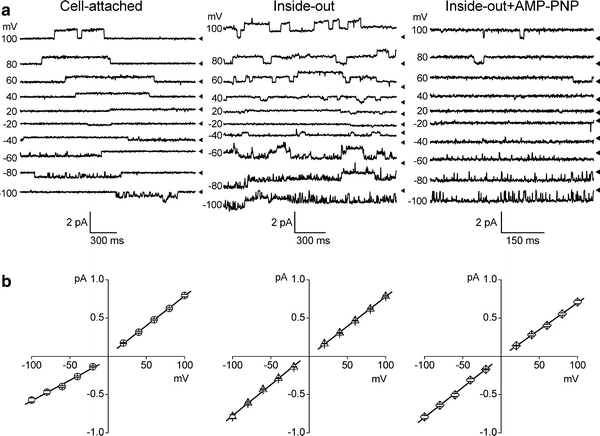
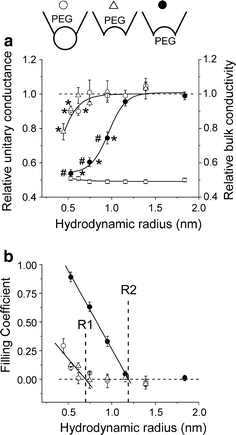
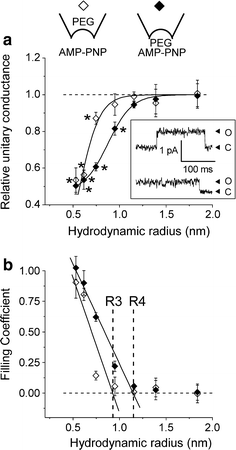
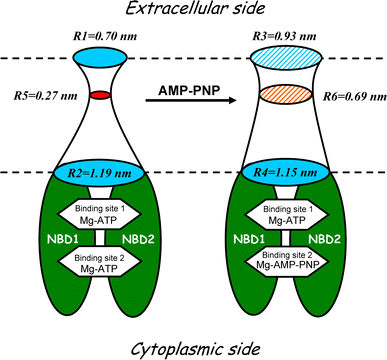
Despite substantial efforts, the entire cystic fibrosis transmembrane conductance regulator (CFTR) protein proved to be difficult for structural analysis at high resolution, and little is still known about the actual dimensions of the anion-transporting pathway of CFTR channel. In the present study, we therefore gauged geometrical features of the CFTR Cl(-) channel pore by a nonelectrolyte exclusion technique. Polyethylene glycols with a hydrodynamic radius (R (h)) smaller than 0.95 nm (PEG 300-1,000) added from the intracellular side greatly suppressed the inward unitary anionic conductance, whereas only molecules with R (h) ≤ 0.62 nm (PEG 200-400) applied extracellularly were able to affect the outward unitary anionic currents. Larger molecules with R (h) = 1.16-1.84 nm (PEG 1,540-3,400) added from either side were completely excluded from the pore and had no significant effect on the single-channel conductance. The cut-off radius of the inner entrance of CFTR channel pore was assessed to be 1.19 ± 0.02 nm. The outer entrance was narrower with its cut-off radius of 0.70 ± 0.16 nm and was dilated to 0.93 ± 0.23 nm when a non-hydrolyzable ATP analog, 5'-adenylylimidodiphosphate (AMP-PNP), was added to the intracellular solution. Thus, it is concluded that the structure of CFTR channel pore is highly asymmetric with a narrower extracellular entrance and that a dilating conformational change of the extracellular entrance is associated with the channel transition to a non-hydrolytic, locked-open state.
Figures

Similar articles
-
A cluster of negative charges at the amino terminal tail of CFTR regulates ATP-dependent channel gating.J Physiol. 2001 Oct 15;536(Pt 2):459-70. doi: 10.1111/j.1469-7793.2001.0459c.xd. J Physiol. 2001. PMID: 11600681 Free PMC article.
-
Comparison of the gating behaviour of human and murine cystic fibrosis transmembrane conductance regulator Cl- channels expressed in mammalian cells.J Physiol. 1998 Apr 15;508 ( Pt 2)(Pt 2):379-92. doi: 10.1111/j.1469-7793.1998.379bq.x. J Physiol. 1998. PMID: 9508803 Free PMC article.
-
ATP hydrolysis cycles and the gating of CFTR Cl- channels.Acta Physiol Scand Suppl. 1998 Aug;643:247-56. Acta Physiol Scand Suppl. 1998. PMID: 9789567 Review.
-
Conserved allosteric hot spots in the transmembrane domains of cystic fibrosis transmembrane conductance regulator (CFTR) channels and multidrug resistance protein (MRP) pumps.J Biol Chem. 2014 Jul 18;289(29):19942-57. doi: 10.1074/jbc.M114.562116. Epub 2014 May 29. J Biol Chem. 2014. PMID: 24876383 Free PMC article.
-
The gating of the CFTR channel.Cell Mol Life Sci. 2017 Jan;74(1):85-92. doi: 10.1007/s00018-016-2390-z. Epub 2016 Oct 1. Cell Mol Life Sci. 2017. PMID: 27696113 Free PMC article. Review.
Cited by
-
Volume-sensitive anion channels mediate osmosensitive glutathione release from rat thymocytes.PLoS One. 2013;8(1):e55646. doi: 10.1371/journal.pone.0055646. Epub 2013 Jan 30. PLoS One. 2013. PMID: 23383255 Free PMC article.
-
ATP Release Channels.Int J Mol Sci. 2018 Mar 11;19(3):808. doi: 10.3390/ijms19030808. Int J Mol Sci. 2018. PMID: 29534490 Free PMC article. Review.
-
Cell Death Induction and Protection by Activation of Ubiquitously Expressed Anion/Cation Channels. Part 1: Roles of VSOR/VRAC in Cell Volume Regulation, Release of Double-Edged Signals and Apoptotic/Necrotic Cell Death.Front Cell Dev Biol. 2021 Jan 12;8:614040. doi: 10.3389/fcell.2020.614040. eCollection 2020. Front Cell Dev Biol. 2021. PMID: 33511120 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

