Transmembrane pores formed by human antimicrobial peptide LL-37
- PMID: 21463582
- PMCID: PMC3072607
- DOI: 10.1016/j.bpj.2011.02.018
Transmembrane pores formed by human antimicrobial peptide LL-37
Abstract
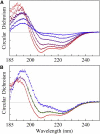
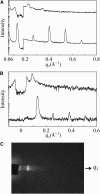
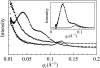
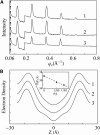
Human LL-37 is a multifunctional cathelicidin peptide that has shown a wide spectrum of antimicrobial activity by permeabilizing microbial membranes similar to other antimicrobial peptides; however, its molecular mechanism has not been clarified. Two independent experiments revealed LL-37 bound to membranes in the α-helical form with the axis lying in the plane of membrane. This led to the conclusion that membrane permeabilization by LL-37 is a nonpore carpet-like mechanism of action. Here we report the detection of transmembrane pores induced by LL-37. The pore formation coincided with LL-37 helices aligning approximately normal to the plane of the membrane. We observed an unusual phenomenon of LL-37 embedded in stacked membranes, which are commonly used in peptide orientation studies. The membrane-bound LL-37 was found in the normal orientation only when the membrane spacing in the multilayers exceeded its fully hydrated value. This was achieved by swelling the stacked membranes with excessive water to a swollen state. The transmembrane pores were detected and investigated in swollen states by means of oriented circular dichroism, neutron in-plane scattering, and x-ray lamellar diffraction. The results are consistent with the effect of LL-37 on giant unilamellar vesicles. The detected pores had a water channel of radius 23-33 Å. The molecular mechanism of pore formation by LL-37 is consistent with the two-state model exhibited by magainin and other small pore-forming peptides. The discovery that peptide-membrane interactions in swollen states are different from those in less hydrated states may have implications for other large membrane-active peptides and proteins studied in stacked membranes.
Copyright © 2011 Biophysical Society. Published by Elsevier Inc. All rights reserved.
Figures


References
-
- Zanetti M. Cathelicidins, multifunctional peptides of the innate immunity. J. Leukoc. Biol. 2004;75:39–48. - PubMed
-
- Scott M.G., Davidson D.J., Hancock R.E. The human antimicrobial peptide LL-37 is a multifunctional modulator of innate immune responses. J. Immunol. 2002;169:3883–3891. - PubMed
-
- Zaiou M. Multifunctional antimicrobial peptides: therapeutic targets in several human diseases. J. Mol. Med. 2007;85:317–329. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

