The calponin regulatory region is intrinsically unstructured: novel insight into actin-calponin and calmodulin-calponin interfaces using NMR spectroscopy
- PMID: 21463585
- PMCID: PMC3072660
- DOI: 10.1016/j.bpj.2011.01.040
The calponin regulatory region is intrinsically unstructured: novel insight into actin-calponin and calmodulin-calponin interfaces using NMR spectroscopy
Abstract
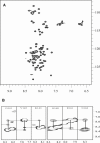
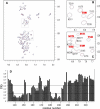
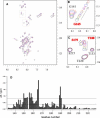
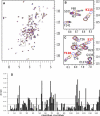
Calponin is an actin- and calmodulin-binding protein believed to regulate the function of actin. Low-resolution studies based on proteolysis established that the recombinant calponin fragment 131-228 contained actin and calmodulin recognition sites but failed to precisely identify the actin-binding determinants. In this study, we used NMR spectroscopy to investigate the structure of this functionally important region of calponin and map its interaction with actin and calmodulin at amino-acid resolution. Our data indicates that the free calponin peptide is largely unstructured in solution, although four short amino-acid stretches corresponding to residues 140-146, 159-165, 189-195, and 199-205 display the propensity to form α-helices. The presence of four sequential transient helices probably provides the conformational malleability needed for the promiscuous nature of this region of calponin. We identified all amino acids involved in actin binding and demonstrated for the first time, to our knowledge, that the N-terminal flanking region of Lys(137)-Tyr(144) is an integral part of the actin-binding site. We have also delineated the second actin-binding site to amino acids Thr(180)-Asp(190). Ca(2+)-calmodulin binding extends beyond the previously identified minimal sequence of 153-163 and includes most amino acids within the stretch 143-165. In addition, we found that calmodulin induces chemical shift perturbations of amino acids 188-190 demonstrating for the first time, to our knowledge, an effect of Ca(2+)-calmodulin on this region. The spatial relationship of the actin and calmodulin contacts as well as the transient α-helical structures within the regulatory region of calponin provides a structural framework for understanding the Ca(2+)-dependent regulation of the actin-calponin interaction by calmodulin.
Copyright © 2011 Biophysical Society. Published by Elsevier Inc. All rights reserved.
Figures


References
-
- Rozenblum G.T., Gimona M. Calponins: adaptable modular regulators of the actin cytoskeleton. Int. J. Biochem. Cell Biol. 2007;40:1990–1995. - PubMed
-
- Takahashi K., Hiwada K., Kokubu T. Isolation and characterization of a 34,000-Dalton calmodulin- and F-actin-binding protein from chicken gizzard smooth muscle. Biochem. Biophys. Res. Commun. 1986;141:20–26. - PubMed
-
- Winder S.J., Walsh M.P. Smooth muscle calponin. Inhibition of actomyosin MgATPase and regulation by phosphorylation. J. Biol. Chem. 1990;265:10148–10155. - PubMed
-
- Applegate D., Feng W., Taubman M.B. Cloning and expression of a novel acidic calponin isoform from rat aortic vascular smooth muscle. J. Biol. Chem. 1994;269:10683–10690. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

