Topical 0.005% tacrolimus eye drop for refractory vernal keratoconjunctivitis
- PMID: 21475312
- PMCID: PMC3178177
- DOI: 10.1038/eye.2011.75
Topical 0.005% tacrolimus eye drop for refractory vernal keratoconjunctivitis
Abstract
Purpose: To evaluate the efficacy and safety of topical 0.005% tacrolimus eye drop for treatment of refractory vernal keratoconjunctivitis (VKC).
Methods: This prospective study included 20 eyes of 10 patients with refractory VKC, who had active symptomatic disease despite conventional medications including topical steroids. After discontinuing all other medications, patients were treated with topical 0.005% tacrolimus eye drop four times a day. Changes in subjective symptoms and objective signs after treatment were evaluated, and development of possible complications was assessed.
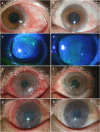
Results: Mean age of patients was 21.3±7.4 years and mean duration of VKC was 12.1±5.8 years. After starting tacrolimus eye drop, patients were followed for a mean duration of 10.7±3.7 months (range, 6-15 months). All symptoms including itching, redness, photosensitivity, foreign body sensation, and mucus discharge improved after the treatment; itching was the first symptom to show dramatic relief. In addition, there was improvement in objective signs including conjunctival hyperaemia, conjunctival papillary hypertrophy, giant papillae, limbal hypertrophy, corneal punctate epithelial erosions, and corneal pannus; conjunctival hyperaemia was the first sign to show improvement. No patient required addition of other medications including steroids for further relief. Any attempt to discontinue tacrolimus eye drop was associated with recurrence of patients' symptoms and signs, necessitating continued use of the medication during the entire follow-up time. No ocular complication related to tacrolimus was noted.
Conclusion: Topical 0.005% tacrolimus eye drop seemed to be a safe and effective treatment for steroid-resistant refractory VKC; however, long-term use was needed to control the disease.
Figures




Similar articles
-
Therapeutic Effect of 0.1% Tacrolimus Eye Drops in the Tarsal Form of Vernal Keratoconjunctivitis.Ophthalmic Res. 2018;59(3):126-134. doi: 10.1159/000478704. Epub 2017 Aug 12. Ophthalmic Res. 2018. PMID: 28803239
-
A large prospective observational study of novel cyclosporine 0.1% aqueous ophthalmic solution in the treatment of severe allergic conjunctivitis.J Ocul Pharmacol Ther. 2009 Aug;25(4):365-72. doi: 10.1089/jop.2008.0103. J Ocul Pharmacol Ther. 2009. PMID: 19441889 Clinical Trial.
-
Tacrolimus in Corticosteroid-Refractory Vernal Keratoconjunctivitis.Cornea. 2016 Nov;35(11):1444-1448. doi: 10.1097/ICO.0000000000000918. Cornea. 2016. PMID: 27310883
-
Use of cyclosporine A and tacrolimus in treatment of vernal keratoconjunctivitis.Curr Allergy Asthma Rep. 2013 Jun;13(3):308-14. doi: 10.1007/s11882-013-0345-0. Curr Allergy Asthma Rep. 2013. PMID: 23625179 Review.
-
Vernal keratoconjunctivitis: a severe allergic eye disease with remodeling changes.Pediatr Allergy Immunol. 2014 Jun;25(4):314-22. doi: 10.1111/pai.12197. Epub 2014 Jan 20. Pediatr Allergy Immunol. 2014. PMID: 24438133 Review.
Cited by
-
Long-term use of 0.003% tacrolimus suspension for treatment of vernal keratoconjunctivitis.Oman J Ophthalmol. 2017 Sep-Dec;10(3):145-149. doi: 10.4103/ojo.OJO_232_2014. Oman J Ophthalmol. 2017. PMID: 29118487 Free PMC article.
-
Treatment of Steroid-Resistant Nodular Episcleritis With Tacrolimus: A Case Report.Cureus. 2023 Oct 15;15(10):e47057. doi: 10.7759/cureus.47057. eCollection 2023 Oct. Cureus. 2023. PMID: 38022063 Free PMC article.
-
Therapeutic effects of topical 0.03% Tacrolimus ointment in children with refractory vernal keratoconjunctivitis in Middle East.Saudi J Ophthalmol. 2019 Apr-Jun;33(2):117-120. doi: 10.1016/j.sjopt.2019.04.001. Epub 2019 May 17. Saudi J Ophthalmol. 2019. PMID: 31384152 Free PMC article.
-
Current Approach to Dry Eye Disease.Clin Rev Allergy Immunol. 2015 Dec;49(3):288-97. doi: 10.1007/s12016-014-8438-7. Clin Rev Allergy Immunol. 2015. PMID: 25081064 Review.
-
Topical Tacrolimus in Anterior Segment Disorders in Ophthalmology: A Review.Rom J Ophthalmol. 2024 Apr-Jun;68(2):92-98. doi: 10.22336/rjo.2024.19. Rom J Ophthalmol. 2024. PMID: 39006333 Free PMC article. Review.
References
-
- Bielory L, Frohman LP. Allergic and immunologic disorders of the eye. J Allergy Clin Immunol. 1992;89 (1 Part 1:1–15. - PubMed
-
- Bonini S, Bonini S, Lambiase A, Marchi S, Pasqualetti P, Zuccaro O, et al. Vernal keratoconjunctivitis revisited: a case series of 195 patients with long-term follow-up. Ophthalmology. 2000;107 (6:1157–1163. - PubMed
-
- Tabbara KF. Ocular complications of vernal keratoconjunctivitis. Can J Ophthalmol. 1999;34 (2:88–92. - PubMed
-
- BenEzra D, Pe'er J, Brodsky M, Cohen E. Cyclosporine eye drops for the treatment of severe vernal keratoconjunctivitis. Am J Ophthalmol. 1986;101 (3:278–282. - PubMed
-
- Secchi AG, Tognon MS, Leonardi A. Topical use of cyclosporine in the treatment of vernal keratoconjunctivitis. Am J Ophthalmol. 1990;110 (6:641–645. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

