Differentiated transplant derived airway epithelial cell cytokine secretion is not regulated by cyclosporine
- PMID: 21477368
- PMCID: PMC3079624
- DOI: 10.1186/1465-9921-12-44
Differentiated transplant derived airway epithelial cell cytokine secretion is not regulated by cyclosporine
Abstract
Background: While lung transplantation is an increasingly utilized therapy for advanced lung diseases, chronic rejection in the form of bronchiolitis obliterans syndrome (BOS) continues to result in significant allograft dysfunction and patient mortality. Despite correlation of clinical events with eventual development of BOS, the causative pathophysiology remains unknown. Airway epithelial cells within the region of inflammation and fibrosis associated with BOS may have a participatory role.
Methods: Transplant derived airway epithelial cells differentiated in air liquid interface culture were treated with IL-1β and/or cyclosporine, after which secretion of cytokines and growth factor and gene expression for markers of epithelial to mesenchymal transition were analyzed.
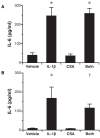
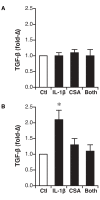
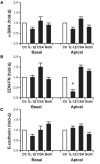
Results: Secretion of IL-6, IL-8, and TNF-α, but not TGF-β1, was increased by IL-1β stimulation. In contrast to previous studies using epithelial cells grown in submersion culture, treatment of differentiated cells in ALI culture with cyclosporine did not elicit cytokine or growth factor secretion, and did not alter IL-6, IL-8, or TNF-α production in response to IL-1β treatment. Neither IL-1β nor cyclosporine elicited expression of markers of the epithelial to mesenchymal transition E-cadherin, EDN-fibronectin, and α-smooth muscle actin.
Conclusion: Transplant derived differentiated airway epithelial cell IL-6, IL-8, and TNF-α secretion is not regulated by cyclosporine in vitro; these cells thus may participate in local inflammatory responses in the setting of immunosuppression. Further, treatment with IL-1β did not elicit gene expression of markers of epithelial to mesenchymal transition. These data present a model of differentiated airway epithelial cells that may be useful in understanding epithelial participation in airway inflammation and allograft rejection in lung transplantation.
Figures



Similar articles
-
Pseudomonas aeruginosa Induced Airway Epithelial Injury Drives Fibroblast Activation: A Mechanism in Chronic Lung Allograft Dysfunction.Am J Transplant. 2016 Jun;16(6):1751-65. doi: 10.1111/ajt.13690. Epub 2016 Feb 26. Am J Transplant. 2016. PMID: 26714197 Free PMC article.
-
TGF-beta1 induced epithelial to mesenchymal transition (EMT) in human bronchial epithelial cells is enhanced by IL-1beta but not abrogated by corticosteroids.Respir Res. 2009 Oct 27;10(1):100. doi: 10.1186/1465-9921-10-100. Respir Res. 2009. PMID: 19857272 Free PMC article.
-
Interleukin-6 and interferon-gamma gene polymorphisms in the development of bronchiolitis obliterans syndrome after lung transplantation.Transplantation. 2002 Nov 15;74(9):1297-302. doi: 10.1097/00007890-200211150-00017. Transplantation. 2002. PMID: 12451269
-
Bronchiolitis obliterans: pathogenesis, prevention, and management.Am J Med Sci. 1998 Mar;315(3):161-78. doi: 10.1097/00000441-199803000-00005. Am J Med Sci. 1998. PMID: 9519929 Review.
-
The contribution of airway ischemia and vascular remodelling to the pathophysiology of bronchiolitis obliterans syndrome and chronic lung allograft dysfunction.Curr Opin Organ Transplant. 2010 Oct;15(5):558-62. doi: 10.1097/MOT.0b013e32833e1112. Curr Opin Organ Transplant. 2010. PMID: 20693899 Review.
Cited by
-
Airway epithelium in lung transplantation: a potential actor for post-transplant complications?Eur Respir Rev. 2024 Nov 27;33(174):240093. doi: 10.1183/16000617.0093-2024. Print 2024 Oct. Eur Respir Rev. 2024. PMID: 39603662 Free PMC article. Review.
-
Adverse effects of immunosuppressant drugs upon airway epithelial cell and mucociliary clearance: implications for lung transplant recipients.Drugs. 2013 Jul;73(11):1157-69. doi: 10.1007/s40265-013-0089-0. Drugs. 2013. PMID: 23842748 Review.
-
Recombinant Acid Ceramidase Reduces Inflammation and Infection in Cystic Fibrosis.Am J Respir Crit Care Med. 2020 Oct 15;202(8):1133-1145. doi: 10.1164/rccm.202001-0180OC. Am J Respir Crit Care Med. 2020. PMID: 32569477 Free PMC article.
References
-
- Christie JD, Edwards LB, Aurora P, Dobbels F, Kirk R, Rahmel AO, Taylor DO, Kucheryavaya AY, Hertz MI. Registry of the International Society for Heart and Lung Transplantation: twenty-fifth official adult lung and heart/lung transplantation report--2008. J Heart Lung Transplant. 2008;27(9):957–969. doi: 10.1016/j.healun.2008.07.018. - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

