Activation of cGMP-dependent protein kinase stimulates cardiac ATP-sensitive potassium channels via a ROS/calmodulin/CaMKII signaling cascade
- PMID: 21479273
- PMCID: PMC3066208
- DOI: 10.1371/journal.pone.0018191
Activation of cGMP-dependent protein kinase stimulates cardiac ATP-sensitive potassium channels via a ROS/calmodulin/CaMKII signaling cascade
Abstract
Background: Cyclic GMP (cGMP)-dependent protein kinase (PKG) is recognized as an important signaling component in diverse cell types. PKG may influence the function of cardiac ATP-sensitive potassium (K(ATP)) channels, an ion channel critical for stress adaptation in the heart; however, the underlying mechanism remains largely unknown. The present study was designed to address this issue.
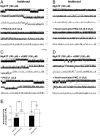
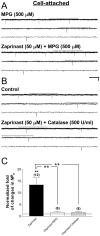
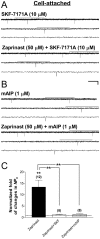
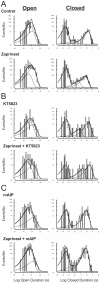
Methods and findings: Single-channel recordings of cardiac K(ATP) channels were performed in both cell-attached and inside-out patch configurations using transfected human embryonic kidney (HEK)293 cells and rabbit ventricular cardiomyocytes. We found that Kir6.2/SUR2A (the cardiac-type K(ATP)) channels were activated by cGMP-selective phosphodiesterase inhibitor zaprinast in a concentration-dependent manner in cell-attached patches obtained from HEK293 cells, an effect mimicked by the membrane-permeable cGMP analog 8-bromo-cGMP whereas abolished by selective PKG inhibitors. Intriguingly, direct application of PKG moderately reduced rather than augmented Kir6.2/SUR2A single-channel currents in excised, inside-out patches. Moreover, PKG stimulation of Kir6.2/SUR2A channels in intact cells was abrogated by ROS/H(2)O(2) scavenging, antagonism of calmodulin, and blockade of calcium/calmodulin-dependent protein kinase II (CaMKII), respectively. Exogenous H(2)O(2) also concentration-dependently stimulated Kir6.2/SUR2A channels in intact cells, and its effect was prevented by inhibition of calmodulin or CaMKII. PKG stimulation of K(ATP) channels was confirmed in intact ventricular cardiomyocytes, which was ROS- and CaMKII-dependent. Kinetically, PKG appeared to stimulate these channels by destabilizing the longest closed state while stabilizing the long open state and facilitating opening transitions.
Conclusion: The present study provides novel evidence that PKG exerts dual regulation of cardiac K(ATP) channels, including marked stimulation resulting from intracellular signaling mediated by ROS (H(2)O(2) in particular), calmodulin and CaMKII, alongside of moderate channel suppression likely mediated by direct PKG phosphorylation of the channel or some closely associated proteins. The novel cGMP/PKG/ROS/calmodulin/CaMKII signaling pathway may regulate cardiomyocyte excitability by opening K(ATP) channels and contribute to cardiac protection against ischemia-reperfusion injury.
Conflict of interest statement
Figures


References
-
- Ashcroft FM. Adenosine 5′-triphosphate-sensitive potassium channels. Annu Rev Neurosci. 1988;11:97–118. - PubMed
-
- Miki T, Seino S. Roles of KATP channels as metabolic sensors in acute metabolic changes. J Mol Cell Cardiol. 2005;38:917–925. - PubMed
-
- Nichols CG. KATP channels as molecular sensors of cellular metabolism. Nature. 2006;440:470–476. - PubMed
-
- Babenko AP, Aguilar-Bryan L, Bryan J. A view of SUR/Kir6.X, KATP channels. Annu Rev Physiol. 1998;60:667–687. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

