Lysosomotropic agents as HCV entry inhibitors
- PMID: 21481279
- PMCID: PMC3090357
- DOI: 10.1186/1743-422X-8-163
Lysosomotropic agents as HCV entry inhibitors
Abstract
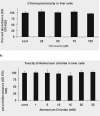
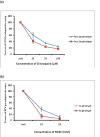
HCV has two envelop proteins named as E1 and E2 which play an important role in cell entry through two main pathways: direct fusion at the plasma membrane and receptor-mediated endocytosis. Fusion of the HCV envelope proteins is triggered by low pH within the endosome. Lysosomotropic agents (LA) such as Chloroquine and Ammonium chloride (NH₄Cl) are the weak bases and penetrate in lysosome as protonated form and increase the intracellular pH. To investigate the antiviral effect of LA (Chloroquine and NH₄Cl) on pH dependent endocytosis, HCV pseudoparticles (HCVpp) of 1a and 3a genotype were produced and used to infect liver cells. The toxicological effects of Chloroquine and NH₄Cl were tested in liver cells through MTT cell proliferation assay. For antiviral screening of Chloroquine and NH₄Cl, liver cells were infected with HCVpp of 3a and 1a genotype in the presence or absence of different concentrations of Chloroquine and NH₄Cl and there luciferase activity was determined by using a luminometer. The results demonstrated that Chloroquine and NH₄Cl showed more than 50% reduction of virus infectivity at 50 μM and 10 mM concentrations respectively. These results suggest that inhibition of HCV at fusion step by increasing the lysosomal pH will be better option to treat chronic HCV.
Figures


References
-
- Nousbaum JB, Pol S, Nalpas B. Group atCS. Hepatitis C virus type 1b (II) infection in France and Italy. Ann Intern Med. 1995;122:161–168. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

