Extended treatment with lamivudine and adefovir dipivoxil in chronic hepatitis B patients with lamivudine resistance
- PMID: 21484148
- PMCID: PMC3090563
- DOI: 10.1007/s12072-010-9228-9
Extended treatment with lamivudine and adefovir dipivoxil in chronic hepatitis B patients with lamivudine resistance
Abstract
Purpose: We and others have reported that adding adefovir dipivoxil (adefovir) to lamivudine results in virological and biochemical improvement in cases of lamivudine resistance. The current study assessed the efficacy and safety of combined therapy after 104 weeks of combined treatment and analyzed the frequency of persistent lamivudine resistant HBV.
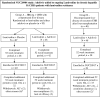
Methods: A total of 78 patients with compensated CHB (Group A) were maintained on either adefovir 10 mg daily (n = 38) or placebo (n = 40) while continuing lamivudine. An additional 38 patients with decompensated cirrhosis or post liver transplantation (Group B) received lamivudine plus adefovir. The primary endpoint was HBV DNA response at year 2.
Results: At week 104 of therapy, a significantly greater proportion of patients in Group A on combination therapy (76%) had a decline in serum HBV DNA to ≤10(5) copies or >2 log(10) reduction from baseline compared to those receiving lamivudine alone (13%; p < 0.001). Fifty-two percent of Group A patients on combination treatment continued to have the M204V/I HBV mutation compared to 92% receiving lamivudine alone (p = 0.0013). Virologic response occurred less frequently in patients expressing persistent lamivudine resistant HBV. In Group B, 87% of patients had HBV DNA response at week 104 (median change from baseline of -5.84 log(10) copies/mL).
Conclusions: The combination of lamivudine and adefovir for 2 years generally proved effective in lamivudine-resistant cases, but there was a persistently high rate of detection of lamivudine resistant mutants and impaired virologic response in compensated patients.
Figures


Similar articles
-
Adefovir dipivoxil added to ongoing lamivudine in chronic hepatitis B with YMDD mutant hepatitis B virus.Gastroenterology. 2004 Jan;126(1):81-90. doi: 10.1053/j.gastro.2003.10.050. Gastroenterology. 2004. PMID: 14699490 Clinical Trial.
-
Adefovir dipivoxil alone or in combination with lamivudine in patients with lamivudine-resistant chronic hepatitis B.Gastroenterology. 2004 Jan;126(1):91-101. doi: 10.1053/j.gastro.2003.10.051. Gastroenterology. 2004. PMID: 14699491 Clinical Trial.
-
Adefovir dipivoxil: a review of its use in chronic hepatitis B.Drugs. 2003;63(20):2215-34. doi: 10.2165/00003495-200363200-00007. Drugs. 2003. PMID: 14498759 Review.
-
Long-term adefovir dipivoxil monotherapy for up to 5 years in lamivudine-resistant chronic hepatitis B.Antivir Ther. 2010;15(2):235-41. doi: 10.3851/IMP1510. Antivir Ther. 2010. PMID: 20386079 Clinical Trial.
-
Treatment of chronic hepatitis B: case selection and duration of therapy.J Gastroenterol Hepatol. 2002 Apr;17(4):409-14. doi: 10.1046/j.1440-1746.2002.02767.x. J Gastroenterol Hepatol. 2002. PMID: 11982721 Review.
Cited by
-
Update on rescue therapies in patients with lamivudine-resistant chronic hepatitis B.Drug Des Devel Ther. 2013 Aug 20;7:777-88. doi: 10.2147/DDDT.S33947. eCollection 2013. Drug Des Devel Ther. 2013. PMID: 23990707 Free PMC article. Review.
-
Efficacy and Safety of Telbivudine Compared to Entecavir Among HBeAg+ Chronic Hepatitis B Patients: a Meta-Analysis Study.Hepat Mon. 2013 May 23;13(6):e7862. doi: 10.5812/hepatmon.7862. eCollection 2013. Hepat Mon. 2013. PMID: 24032045 Free PMC article.
-
Adverse events of nucleos(t)ide analogues for chronic hepatitis B: a systematic review.J Gastroenterol. 2020 May;55(5):496-514. doi: 10.1007/s00535-020-01680-0. Epub 2020 Mar 17. J Gastroenterol. 2020. PMID: 32185517 Free PMC article.
-
Entecavir and hepatitis B immune globulin in patients undergoing liver transplantation for chronic hepatitis B.Liver Transpl. 2013 Aug;19(8):887-95. doi: 10.1002/lt.23690. Liver Transpl. 2013. PMID: 23788462 Free PMC article. Clinical Trial.
-
Two rescue therapies in lamivudine-resistant patients with chronic hepatitis B in the central China: adefovir monotherapy and adefovir plus lamivudine.Virus Genes. 2014 Feb;48(1):32-7. doi: 10.1007/s11262-013-1004-1. Epub 2013 Nov 8. Virus Genes. 2014. PMID: 24203098
References
-
- Schalm SW, Heathcote J, Cianciara J, Farrell G, Sherman M, Willems B, Dhillon A, Moorat A, Barber J, Gray DF. Lamivudine and alpha interferon combination treatment of patients with chronic hepatitis B infection: a randomized trial. Gut. 2000;46:562–568. doi: 10.1136/gut.46.4.562. - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources

