The Cav3.2 T-type calcium channel regulates temporal coding in mouse mechanoreceptors
- PMID: 21486775
- PMCID: PMC3098700
- DOI: 10.1113/jphysiol.2010.203463
The Cav3.2 T-type calcium channel regulates temporal coding in mouse mechanoreceptors
Abstract
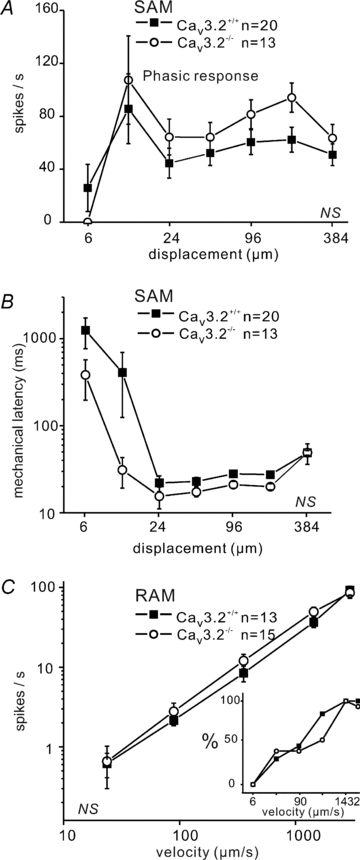
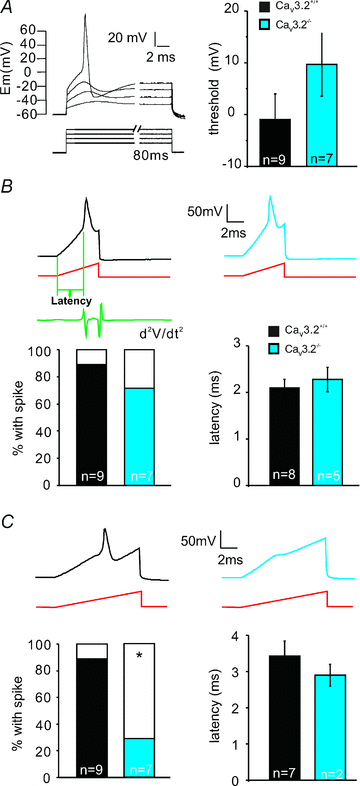
In mammals there are three types of low-voltage-activated (LVA) calcium channels,Cav3.1, Cav3.2 and Cav3.3, which all give rise to T-type Ca2+currents. T-type Ca2+currents have long been known to be highly enriched in a sub-population of medium-sized sensory neurones in the dorsal root ganglia (DRG). However, the identity of the T-type-rich sensory neurones has remained controversial and the precise physiological role of the Cav3.2 calcium channel in these sensory neurones has not been directly addressed. Here we show, using Cav3.2−/− mutant mice,that these channels are essential for the normal temporal coding of moving stimuli by specialized skin mechanoreceptors called D-hair receptors.We show that D-hair receptors from Cav3.2−/− fire approximately 50% fewer spikes in response to ramp-and-hold displacement stimuli compared to wild type receptors. The reduced sensitivity of D-hair receptors in Cav3.2−/− mice is chiefly due to an increase in the mechanical threshold and a substantial temporal delay in the onset of high-frequency firing to moving stimuli.We examined the receptive properties of other cutaneous mechano receptors and Aδ- and C-fibre nociceptors in Cav3.2−/− mice, but found no alteration in their mechanosensitivity compared to Cav3.2+/+mice. However, C-fibre nociceptors recorded in Cav3.2−/− mutant mice displayed a small but statistically significant reduction in their spiking rate during noxious heat ramps when compared to C-fibres in control mice. The T-type calcium channel Cav3.2 is thus not only a highly specific marker of D-hair receptors but is also required to maintain their high sensitivity and above all to ensure ultra rapid temporal detection of skin movement.
Figures


Comment in
-
Mice mechanoreceptors are modulated by Ca(v)3.2 T-type calcium channels.J Physiol. 2012 Jan 1;590(1):7. doi: 10.1113/jphysiol.2011.221507. J Physiol. 2012. PMID: 22210285 Free PMC article. No abstract available.
References
-
- Airaksinen MS, Koltzenburg M, Lewin GR, Masu Y, Helbig C, Wolf E, Brem G, et al. Specific subtypes of cutaneous mechanoreceptors require neurotrophin-3 following peripheral target innervation. Neuron. 1996;16:287–295. - PubMed
-
- Aptel H, Hilaire C, Pieraut S, Boukhaddaoui H, Mallie S, Valmier J, Scamps F. The Cav3.2/α1H T-type Ca2+ current is a molecular determinant of excitatory effects of GABA in adult sensory neurons. Mol Cell Neurosci. 2007;36:293–303. - PubMed
-
- Barton ME, Eberle EL, Shannon HE. The antihyperalgesic effects of the T-type calcium channel blockers ethosuximide, trimethadione, and mibefradil. Eur J Pharmacol. 2005;521:79–85. - PubMed
-
- Bilici D, Akpinar E, Gursan N, Dengiz GO, Bilici S, Altas S. Protective effect of T-type calcium channel blocker in histamine-induced paw inflammation in rat. Pharmacol Res. 2001;44:527–531. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

