Adenosine A2A receptor induces protein kinase A-dependent functional modulation of human (alpha)3(beta)4 nicotinic receptor
- PMID: 21486776
- PMCID: PMC3112553
- DOI: 10.1113/jphysiol.2011.207282
Adenosine A2A receptor induces protein kinase A-dependent functional modulation of human (alpha)3(beta)4 nicotinic receptor
Abstract
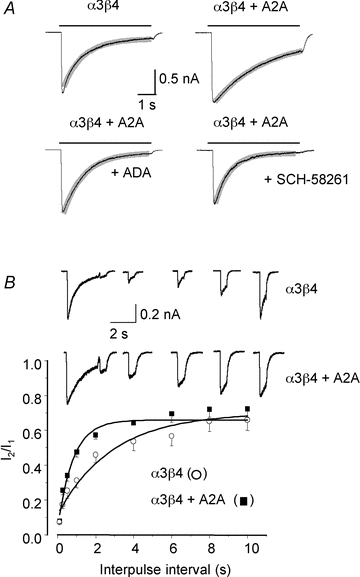
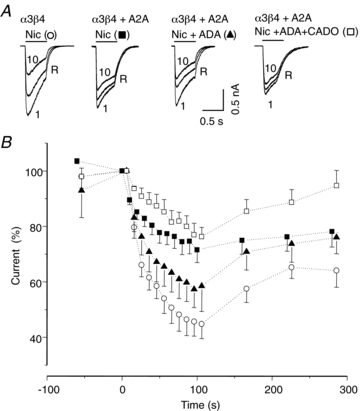
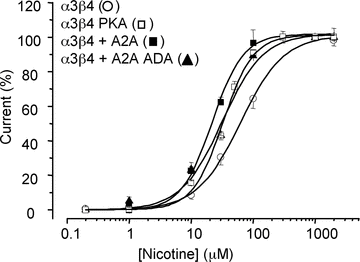
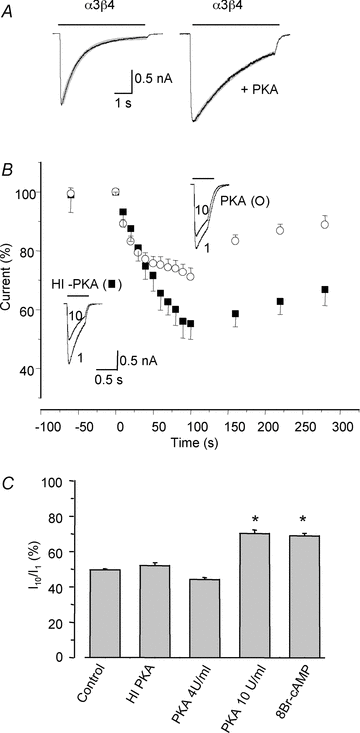
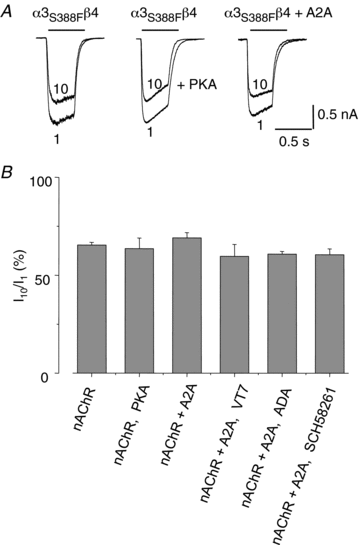
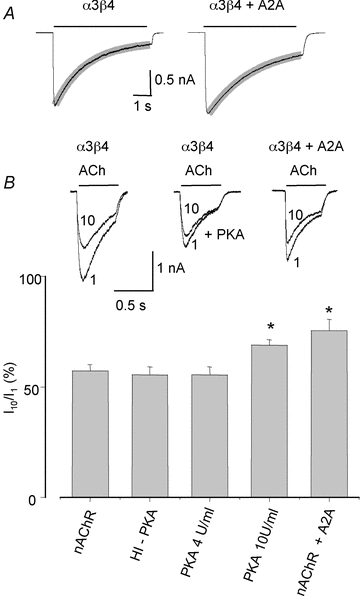
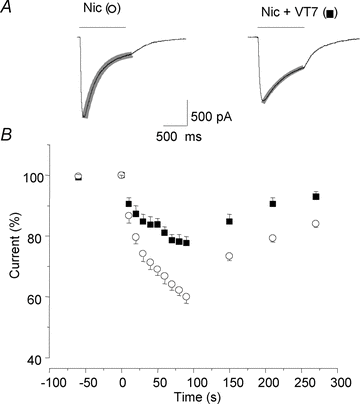
Adenosine modulates the function of nicotinic ACh receptors (nAChRs) in a variety of preparations, possibly through pathways involving protein kinase A (PKA), but these phenomena have not yet been investigated in detail. In this work we studied, using the patch clamp technique, the functional modulation of recombinant human α3β4 nAChR by the A2A adenosine receptor, co-expressed in HEK cells. Tonic activation of A2A receptor slowed current decay during prolonged applications of nicotine and accelerated receptor recovery from desensitization. Together, these changes resulted into a more sustained current response upon multiple nicotine or ACh applications. These findings were confirmed in cultured mouse superior cervical ganglion neurones, which express nAChR containing the α3 subunit together with β2 and/or β4 and A2A receptor. Expression of the A2A receptor in HEK cells also increased the apparent potency of nAChR for nicotine, further supporting a general A2A-induced gain of function for nAChR. These effects were dependent on PKA since the direct activation of PKA mimicked, and its inhibition prevented almost completely, the effects of the A2A receptor. Mutations of R385 and S388 in the cytoplasmic loop of the α3 subunit abolished the functional modulation of nAChR induced by activation of A2A receptor, PKA and other Ser/Thr kinases, suggesting that this region constitutes a putative consensus site for these kinases. These data provide conclusive evidence that activation of the A2A receptor determines functional changes
Figures
References
-
- Caratsch CG, Grassi F, Eusebi F. Functional regulation of nicotinic acetylcholine receptor channels in muscle. Ion Channels. 1992;3:177–206. - PubMed
-
- Correia-de-Sá P, Ribeiro JA. Tonic adenosine A2A receptor activation modulates nicotinic autoreceptor function at the rat neuromuscular junction. Eur J Pharmacol. 1994;271:349–355. - PubMed
-
- Dani JA. Overview of nicotinic receptors and their roles in the central nervous system. Biol Psychiatry. 2001;49:166–174. - PubMed
-
- Di Angelantonio S, De Stefano ME, Piccioni A, Lombardi L, Gotti C, Paggi P. Lack of dystrophin functionally affectsα3β2/β4-nicotinic acethylcholine receptors in sympathetic neurons of dystrophic mdx mice. Neurobiol Dis. 2011;41:528–537. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

